Volume 12, Issue 2 (June 2025)
J. Food Qual. Hazards Control 2025, 12(2): 94-103 |
Back to browse issues page
Ethics code: Not applicable.
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Khatun T, Asaduzzaman M. The Design of Food Safety Management System (FSMS) Plan for Rice Fortification Kernel (RFK) Factory – A Case Study. J. Food Qual. Hazards Control 2025; 12 (2) :94-103
URL: http://jfqhc.ssu.ac.ir/article-1-1282-en.html
URL: http://jfqhc.ssu.ac.ir/article-1-1282-en.html
Department of Food Engineering and Nutrition Science, State University of Bangladesh, State Key Laboratory of Food Science and Resources, Jiangnan University, Wuxi 214122, China, School of Food Science and Technology, Jiangnan University, 1800 Lihu Avenue, 214122 Wuxi, Jiangsu Province, China , bitatipu@gmail.com
Full-Text [PDF 558 kb]
(368 Downloads)
| Abstract (HTML) (867 Views)
Table 1: Product description of Fortified Rice Kernels (FRKs)
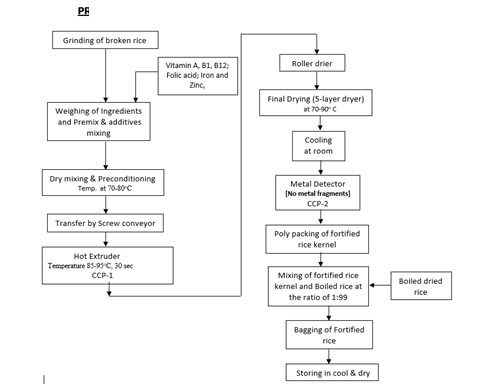
Figure 1: Process flow diagram of Fortified Rice Kernels (FRKs)
Table 2: Hazards in operations, ingredients, and raw material investigation chart
Table 3: Hazards analysis chart for Fortified Rice Kernels (FRKs) processing step
Table 6: Food Safety Management System (FSMS) control chart for rice fortification production
CCP=Critical Control Point; MSDS=Material Safety Data Sheet; QC/QA=Quality Assurance and Quality Control
Full-Text: (262 Views)
The Design of Food Safety Management System (FSMS) Plan for Rice Fortification Kernel (RFK) Factory – A Case Study
T. Khatun 1, M. Asaduzzaman 2,3,4[*]*
1. Department of Mathematics, Chittagong College, National University, Bangladesh
2. Department of Food Engineering and Nutrition Science, State University of Bangladesh
3. State Key Laboratory of Food Science and Resources, Jiangnan University, Wuxi 214122, China
4. School of Food Science and Technology, Jiangnan University, 1800 Lihu Avenue, 214122 Wuxi, Jiangsu Province, China
T. Khatun 1, M. Asaduzzaman 2,3,4[*]*
1. Department of Mathematics, Chittagong College, National University, Bangladesh
2. Department of Food Engineering and Nutrition Science, State University of Bangladesh
3. State Key Laboratory of Food Science and Resources, Jiangnan University, Wuxi 214122, China
4. School of Food Science and Technology, Jiangnan University, 1800 Lihu Avenue, 214122 Wuxi, Jiangsu Province, China
HIGHLIGHTS
- This study investigated the raw materials used for fortification and found two possible processing hazards.
- It proposed possible mitigation strategies for these hazards during factory operations.
- Statistical analysis was not conducted, as the study aimed to present conceptual insights based on observations from various factory settings.
| Article type Original article |
ABSTRACT Background: The Food Safety Management System (FSMS) functions as a protective mechanism in food manufacturing to ensure consumer safety. In contemporary times, it has evolved into a crucial tool for managing various types of food products. The objective of this research was to formulate a precise FSMS plan tailored to the Bangladeshi Rice Fortification Kernel manufacturing factory in Brahmanbaria, Chittagong. Methods: A detailed FSMS model was built to enhance the quality and safety of the Rice Fortification Kernel produced in this factory. This model was designed based on the current circumstances of the rice fortification manufacturing factory, incorporating FSMS's three principles and various existing generic models such as Bangladesh Standards and Testing Institution, Halal certification, the Global Food Safety Initiative, and YUM's Quality Systems Audit of FSMS. Both qualitative and quantitative approaches were utilized. An eight-member FSMS team was established to implement and develop the Hazard Analysis and Critical Control Points system, including validation and control measures. Results: This factory's production of rice fortification identified two Critical Control Points. These points focused on maintaining the proper extruder temperature and implementing metal detection to identify foreign and unwanted materials in the final product. Conclusion: The study's FSMS strategy was instantly applied to the kernel production process of rice fortification. It is broadly recommended that FSMS systems be adopted across all fortification factories to ensure comprehensive food safety for the public. © 2025, Shahid Sadoughi University of Medical Sciences. This is an open access article under the Creative Commons Attribution 4.0 International License. |
|
| Keywords Oryza Hazard Analysis and Critical Control Points Food Safety Bangladesh |
||
| Article history Received: 11 Dec 2024 Revised: 10 Mar 2025 Accepted: 18 June 2025 |
||
| Abbreviations CCP=Critical Control Point FSMS=Food Safety Management System HACCP=Hazard Analysis and Critical Control Point ISO=International Organization for Standardization RFK=Rice Fortification Kernel WFP=World Food Programme |
To cite: Khatun T., Asaduzzaman M. (2025). The design of food safety management system (FSMS) plan for rice fortification kernel (RFK) factory – a case study. Journal of Food Quality and Hazards Control. 12: 94-103.
Introduction
Introduction
Rice fortification is a nutrient-rich, vitamin, and mineral-enriched, easily transportable, long-lasting, and effortlessly storable food product. Possessing a typically robust and dry composition, it thrives in optimal temperatures and endures rigorous handling. Its longevity, without spoilage, spans several years, contingent upon maintaining dryness and the ideal temperature (Thakur et al., 2025). Derived from white, parboiled, or broken rice, rice fortification incorporates a vitamin premix comprising vitamins A (palmitate), B1 (thiamin mononitrate), B12 (cyanocobalamine), folic acid (Vitamin B9), iron (ferric pyrophosphate), and zinc (zinc oxide) (BSTI, 2015). These Fortified Rice Kernels (FRKs), enriched with essential vitamins and minerals, serve as viable components for emergency feeding programs, addressing issues like malnutrition and supporting various health promotion initiatives. Nevertheless, the acceptance of rice fortification hinges on its macro-nutritional, micro-nutritional, and organoleptic attributes, with production costs playing a pivotal role.
The Food Safety Management System (FSMS) is now a tangible and specific method of ensuring food safety from primary production to ultimate consumption in a "farm-to-table" system. ISO 22000, which was developed in the mid-2000s by the International Organization for Standardization (ISO) in collaboration with Good Standardization Practices (GSP) and the World Trade Organization (WTO), was developed to ensure food safety and preservation for all consumer groups and not merely to react to microbiological risks (Asaduzzaman, 2021). FSMS is a preventative system that goes beyond normal quality control or inspection systems. It not only ensures a "zero-risk" atmosphere but also decreases the likelihood of hazards entering food products by principles such as interactive communication, system management, prerequisite programs, and Hazard Analysis and Critical Control Points (HACCP). The optimal method in ensuring food safety is the focus on preventing and controlling probable hazards and the optimization of the production process through Plan-Do-Check-Act (PDCA) cycles (De Lima et al., 2025; De Oleo et al., 2024).
The FSMS is a methodical approach that serves as a tool to ensure food safety in the modern age. Its primary objective is to guarantee the prevention of all food-borne hazards, such as chemical, physical, or biological hazards, from the production stage up to packaging and distribution. The fundamental objectives of any food manufacturing factory are the assurance of proper production and the supply of a variety of safe and healthy food products. Achieving these goals requires an organized and organizational strategy, coordinating actions, procedures, monitoring, and resources, according to standards that sustain total quality systems and HACCP. This includes adherence to ISO 9000:2015 and ISO 22000 series, as well as the FSMS (Rincon-Ballesteros et al., 2024). Fundamental prerequisites for FSMS implementation encompass human resources and financial safety. Initial inputs involve staff training, machinery acquisition, and other technical support to establish a robust FSMS framework (Oruc et al., 2024).
In Bangladesh, mothers and babies mostly suffer from malnutrition, as identified by both the World Food Programme (WFP) and the government of Bangladesh. They mainly suffer from deficiency of the four main vitamins, vitamin A, B1, B9, B12, and two related minerals, which are iron (Fe) and zinc (Zn). Firstly, to minimize and eventually resolve this problem, the Bangladesh government is taking the initiative to establish rice fortification factories through local investors, with facilitation from the WFP, a United Nations organization. The primary requirement of the WFP is to establish FSMS in the factories to help achieve ISO 9001:2015 and ISO 22000 certification, whose ultimate goal is providing safe product for the consumers.
In smaller organizations, particularly those that are family-owned or owned and managed, a higher level of managerial commitment and employee involvement is typically apparent. This is perhaps due to the shared attention to basics such as product flavor, nutritional value, quality, and safety. Additionally, the proximity among employees and customers in these centers can encourage a higher level of personal responsibility and accountability (De Boeck et al., 2016).
FSMS has recently emerged in the factory environment with the intention to handle the human element of food safety law and regulation. In this model, food safety culture within an organization has a significant influence on employees' behavior, both in the manner in which the procedures are executed and in the decision-making process regarding food safety. Contrary to formal systems and procedures, food safety practices are less formal and the outcomes of individual employee perceptions and attitudes. These perceptions, in turn, over time develop a stable belief system and attitudes that persist in the organization (Dzwolak, 2019).
Food safety culture is thus described as the dynamic interplay between employees' food safety perceptions and the organizational environment, in which the FSMS is implemented. This system comprises both control and assurance roles and finally determines the safety and hygiene of food products, the processing environment, and worker hygiene. The FSMS reflects the general beliefs of employees towards leadership, communication, organizational commitment, availability of resources, and perception of risks in relation to food safety and hygiene performance. To make this cultural element easily assessable and improvable, an authentic self-assessment tool has been constructed to measure food safety climate for food businesses (De Boeck et al., 2016; Dzwolak, 2019).
The primary aim of this study was to thoroughly examine the HACCP plans and related documentation within standardized FSMSs implemented in rice fortification facilities. The study focused on two main goals: first, to identify and prioritize hazards relevant to the development of HACCP plans and associated documentation; second, to assess and rank instances of non-compliance and pinpoint hazards or weaknesses within the HACCP systems to establish FSMS.
The Food Safety Management System (FSMS) is now a tangible and specific method of ensuring food safety from primary production to ultimate consumption in a "farm-to-table" system. ISO 22000, which was developed in the mid-2000s by the International Organization for Standardization (ISO) in collaboration with Good Standardization Practices (GSP) and the World Trade Organization (WTO), was developed to ensure food safety and preservation for all consumer groups and not merely to react to microbiological risks (Asaduzzaman, 2021). FSMS is a preventative system that goes beyond normal quality control or inspection systems. It not only ensures a "zero-risk" atmosphere but also decreases the likelihood of hazards entering food products by principles such as interactive communication, system management, prerequisite programs, and Hazard Analysis and Critical Control Points (HACCP). The optimal method in ensuring food safety is the focus on preventing and controlling probable hazards and the optimization of the production process through Plan-Do-Check-Act (PDCA) cycles (De Lima et al., 2025; De Oleo et al., 2024).
The FSMS is a methodical approach that serves as a tool to ensure food safety in the modern age. Its primary objective is to guarantee the prevention of all food-borne hazards, such as chemical, physical, or biological hazards, from the production stage up to packaging and distribution. The fundamental objectives of any food manufacturing factory are the assurance of proper production and the supply of a variety of safe and healthy food products. Achieving these goals requires an organized and organizational strategy, coordinating actions, procedures, monitoring, and resources, according to standards that sustain total quality systems and HACCP. This includes adherence to ISO 9000:2015 and ISO 22000 series, as well as the FSMS (Rincon-Ballesteros et al., 2024). Fundamental prerequisites for FSMS implementation encompass human resources and financial safety. Initial inputs involve staff training, machinery acquisition, and other technical support to establish a robust FSMS framework (Oruc et al., 2024).
In Bangladesh, mothers and babies mostly suffer from malnutrition, as identified by both the World Food Programme (WFP) and the government of Bangladesh. They mainly suffer from deficiency of the four main vitamins, vitamin A, B1, B9, B12, and two related minerals, which are iron (Fe) and zinc (Zn). Firstly, to minimize and eventually resolve this problem, the Bangladesh government is taking the initiative to establish rice fortification factories through local investors, with facilitation from the WFP, a United Nations organization. The primary requirement of the WFP is to establish FSMS in the factories to help achieve ISO 9001:2015 and ISO 22000 certification, whose ultimate goal is providing safe product for the consumers.
In smaller organizations, particularly those that are family-owned or owned and managed, a higher level of managerial commitment and employee involvement is typically apparent. This is perhaps due to the shared attention to basics such as product flavor, nutritional value, quality, and safety. Additionally, the proximity among employees and customers in these centers can encourage a higher level of personal responsibility and accountability (De Boeck et al., 2016).
FSMS has recently emerged in the factory environment with the intention to handle the human element of food safety law and regulation. In this model, food safety culture within an organization has a significant influence on employees' behavior, both in the manner in which the procedures are executed and in the decision-making process regarding food safety. Contrary to formal systems and procedures, food safety practices are less formal and the outcomes of individual employee perceptions and attitudes. These perceptions, in turn, over time develop a stable belief system and attitudes that persist in the organization (Dzwolak, 2019).
Food safety culture is thus described as the dynamic interplay between employees' food safety perceptions and the organizational environment, in which the FSMS is implemented. This system comprises both control and assurance roles and finally determines the safety and hygiene of food products, the processing environment, and worker hygiene. The FSMS reflects the general beliefs of employees towards leadership, communication, organizational commitment, availability of resources, and perception of risks in relation to food safety and hygiene performance. To make this cultural element easily assessable and improvable, an authentic self-assessment tool has been constructed to measure food safety climate for food businesses (De Boeck et al., 2016; Dzwolak, 2019).
The primary aim of this study was to thoroughly examine the HACCP plans and related documentation within standardized FSMSs implemented in rice fortification facilities. The study focused on two main goals: first, to identify and prioritize hazards relevant to the development of HACCP plans and associated documentation; second, to assess and rank instances of non-compliance and pinpoint hazards or weaknesses within the HACCP systems to establish FSMS.
Materials and methods
Study area
This study was performed at a Rice Fortification Kernel (RFK) factory in Brahmanbaria, Chittagong division, Bangladesh. This study focused on raw materials sourced from local markets in the Chittagong and Dhaka divisions. Data collection involved monitoring and verifying various manufacturing stages, from raw material reception to processing, storage, and finished product distribution, encompassing all prevailing procedures. Supplementary insights were derived from factory management and staff, as well as analytical records from the Bangladesh Standards and Testing Institution (BSTI) food processing and engineering laboratory.
Product description
The product description provides a comprehensive overview of various safety aspects, encompassing composition, physical and chemical structure, microbial treatment, packing materials, shelf-life, storage conditions for raw, and finished products, distribution methods, and the material safety data sheet. Additionally, it outlines the intended use of ingredients (Asaduzzaman, 2021). A detailed product description for rice fortification is presented in Table 1.
Study area
This study was performed at a Rice Fortification Kernel (RFK) factory in Brahmanbaria, Chittagong division, Bangladesh. This study focused on raw materials sourced from local markets in the Chittagong and Dhaka divisions. Data collection involved monitoring and verifying various manufacturing stages, from raw material reception to processing, storage, and finished product distribution, encompassing all prevailing procedures. Supplementary insights were derived from factory management and staff, as well as analytical records from the Bangladesh Standards and Testing Institution (BSTI) food processing and engineering laboratory.
Product description
The product description provides a comprehensive overview of various safety aspects, encompassing composition, physical and chemical structure, microbial treatment, packing materials, shelf-life, storage conditions for raw, and finished products, distribution methods, and the material safety data sheet. Additionally, it outlines the intended use of ingredients (Asaduzzaman, 2021). A detailed product description for rice fortification is presented in Table 1.
Table 1: Product description of Fortified Rice Kernels (FRKs)
| 01 | Product name | FRK |
| 02 | Product description | Rice kernels prepared from white or parboiled, or broken rice, and a vitamin premix. |
| 03 | How is it to be used? | Boiled to eat |
| 04 | Packaging | 3-layer craft paper compatible to food grade |
| 05 | Shelf-life | One year |
| 06 | Selling places | Mega shop, retail local store |
| 07 | Labeling and packaging instructions | Optimum temperature |
| 08 | Delivery circumstance | Optimum temperature |
Research method
Employing a qualitative approach, the research provided in-depth insights into the program's current state. The observation extended to the behavior of management review committee members and factory staff, including their interactions, employee relationships, and organizational dynamics. This qualitative method facilitated an exploration of intricate details that might be challenging to capture through quantitative research. As such, the investigative and open-minded nature of this qualitative research aligns with the study's objectives (Asaduzzaman, 2021).
Study approach
The researchers meticulously devised a detailed FSMS plan tailored to the context and operations of the rice fortification factory, aiming to enhance the quality of FRKs. Drawing upon FSMS principles and various established models, this study formulated documentation and recordkeeping forms. These included elements such as the Operational Prerequisite Programme (OPRP) and Prerequisite Programme (PRP); details on the fortification factory’s location, performance, premises, production floor, and equipment; through product descriptions including nutritional components, lists of product apparatuses, machinery, and incoming additives, raw materials; detailed process flow diagrams, traceability, and validation; hazard identification and description; Critical Control Point’s (CCPs) establishment, and a hazard control, and mechanism chart (Nandeep et al., 2024).
Results and discussion
Prerequisite programs
This study encompassed all prerequisite programs within the FSMS, aligning them towards a shared objective of zero defects and preventing health hazards in the final products. The factory implemented various quality control and assurance programs grounded in food production personnel hygiene, Good Manufacturing Practices (GMPs), and Total Quality Management (TQM) (Okpala and Korzeniowska, 2023).
Location
In Brahmanbaria, the factory mitigates food safety risks by avoiding environmentally contaminated areas and industrial activities. Additionally, it boasts safeguards against flooding and natural disasters, making it impervious to pest infestations.
Design and layout of the factory
The factory infrastructure is designed with a focus on hygiene and safety. The absence of slopes in the building and store area, along with well-drained surfaces, eliminates stagnant water, waste, and debris. Adhering to GMPs prevents cross-contamination at every processing stage. The walls of both the building and storehouse are water and pest-proof, with sealed angles, corners, and junctions. The production floor is surrounded by a covered drain connected to a wastewater factory, ensuring efficient liquid drainage. Netted glass doors with automatic closing mechanisms, along with exhaust fans, facilitate fresh airflow, maintaining optimal temperature, and humidity. Epoxy-painted floors and walls simplify sanitation.
The factory prioritizes lighting and ventilation, utilizing glass doors, and windows with aluminum structures. Regular cleaning and maintenance by professional technicians contribute to a pristine environment. Hand washing stations are strategically located near processing, handling, and resting areas, minimizing the risk of cross-contamination. A dedicated quality control laboratory conducts essential experiments, such as proximate analysis and microbial tests on rice kernels, covering both raw materials and final products. This comprehensive approach ensures that the factory adheres to rigorous cleanliness and safety standards, contributing to the production of high-quality FRKs.
Equipment
The equipment, constructed from stainless steel, plastic, and other food-grade materials, features an easily maintainable design. Regular semi-annual preventive maintenance guarantees an efficient, crack-free, rust-free, and dent-free operational system.
Personal hygiene
Adherence to good manufacturing practices ensures the establishment of robust personal hygiene standards among employees, as per the Standard Operating Procedure (SOP). Employees strictly comply with personal hygiene instructions, donning uniforms, gloves, and hairnets to prevent any potential cross-contamination. The guidelines explicitly prohibit actions such as placing fingers in sensitive areas like the mouth, ears, eyes, and nose. Additionally, activities like eating, spitting, chewing, drinking, and smoking are strictly forbidden during food equipment and materials handling and operations. A committed quality control supervisor ensures these practices are followed and keeps records verifying that employees wash and sanitize their hands before starting work and after returning from the washrooms.
Water supply
The factory features a dedicated underground water supply and storage system, delivering sufficient clean, and drinking water post reverse osmosis treatment. This portable, treated water undergoes hourly testing and adheres to Bangladesh’s national water quality standards.
Storage and transportation
Every storage room, except the maintenance store, is meticulously cleaned, sanitized, and maintained with controlled temperature and humidity, featuring air conditioning. The maintenance store is monitored and verified using a hygrometer and data logger. Regular inspections ensure a consistent environment, mitigating hazards, and facilitating the production of finished products, which maintain the same quality from the production to the consumer end. The factory is equipped with proper transportation facilities, overseeing temperature, and separation through the transit of both types of materials, such as raw and finished goods, to ensure compliance with food safety standards.
Sanitation program
Maintaining a sanitary and culturally approachable atmosphere is crucial for storing any kind of goods to ensure providing a safe product for the consumer.
Pest control management
Pest control in the factory aims to remove various kinds and sizes of pests, including human-made incidents. Modern pest management programs, surpassing traditional insecticide spraying and trapping techniques, are employed monthly by factory employees and contracted third-party service providers.
Waste management
The company operates its waste treatment factory with a comprehensive drainage, storage, and disposal system. It consistently checks and tests incoming waste and discharging wastewater to ensure compliance with international standards.
Traceability
Accurate record-keeping includes lot identification numbers, batch numbers, and key dates such as the arrival of raw materials and vitamin premix, production, and premix making. Strict adherence to the First In, First Out (FIFO) system is maintained in the store warehouse for both raw and packaging materials.
Training
Quality control officers conduct bi-weekly training sessions for all employees, addressing essential aspects such as maintaining personal and operational hygiene, operational and handling safety, organizational law, policy and regulations, production processes, and food safety and distribution issues.
Production process
The incoming white, parboiled, or broken rice undergoes in-house crushing using a grinder. The resulting powder is weighed and blended with vitamin premix such as palmitate or acetate as Vitamin A, thiamine mononitrate as vitamin B1, cyanocobalamin as B12, folic acid as B9, ferric pyrophosphate as Fe, and zinc oxide as Zn. The dry mixture undergoes preconditioning in a preconditioned at 70 to 80 °C, then moves to a screw conveyor. A hot extruder, maintained at 85 to 95 °C for 30 sec, produces FRKs. The FRK is subsequently transferred to a roller drier to eliminate excess moisture, followed by a multi-layer dryer at 70 to 90 °C. After cooling and metal detection, the FRK is conveyed to the packaging room, utilizing a food-grade three-layer craft paper. The process flow is illustrated in Figure 1.
Employing a qualitative approach, the research provided in-depth insights into the program's current state. The observation extended to the behavior of management review committee members and factory staff, including their interactions, employee relationships, and organizational dynamics. This qualitative method facilitated an exploration of intricate details that might be challenging to capture through quantitative research. As such, the investigative and open-minded nature of this qualitative research aligns with the study's objectives (Asaduzzaman, 2021).
Study approach
The researchers meticulously devised a detailed FSMS plan tailored to the context and operations of the rice fortification factory, aiming to enhance the quality of FRKs. Drawing upon FSMS principles and various established models, this study formulated documentation and recordkeeping forms. These included elements such as the Operational Prerequisite Programme (OPRP) and Prerequisite Programme (PRP); details on the fortification factory’s location, performance, premises, production floor, and equipment; through product descriptions including nutritional components, lists of product apparatuses, machinery, and incoming additives, raw materials; detailed process flow diagrams, traceability, and validation; hazard identification and description; Critical Control Point’s (CCPs) establishment, and a hazard control, and mechanism chart (Nandeep et al., 2024).
Results and discussion
Prerequisite programs
This study encompassed all prerequisite programs within the FSMS, aligning them towards a shared objective of zero defects and preventing health hazards in the final products. The factory implemented various quality control and assurance programs grounded in food production personnel hygiene, Good Manufacturing Practices (GMPs), and Total Quality Management (TQM) (Okpala and Korzeniowska, 2023).
Location
In Brahmanbaria, the factory mitigates food safety risks by avoiding environmentally contaminated areas and industrial activities. Additionally, it boasts safeguards against flooding and natural disasters, making it impervious to pest infestations.
Design and layout of the factory
The factory infrastructure is designed with a focus on hygiene and safety. The absence of slopes in the building and store area, along with well-drained surfaces, eliminates stagnant water, waste, and debris. Adhering to GMPs prevents cross-contamination at every processing stage. The walls of both the building and storehouse are water and pest-proof, with sealed angles, corners, and junctions. The production floor is surrounded by a covered drain connected to a wastewater factory, ensuring efficient liquid drainage. Netted glass doors with automatic closing mechanisms, along with exhaust fans, facilitate fresh airflow, maintaining optimal temperature, and humidity. Epoxy-painted floors and walls simplify sanitation.
The factory prioritizes lighting and ventilation, utilizing glass doors, and windows with aluminum structures. Regular cleaning and maintenance by professional technicians contribute to a pristine environment. Hand washing stations are strategically located near processing, handling, and resting areas, minimizing the risk of cross-contamination. A dedicated quality control laboratory conducts essential experiments, such as proximate analysis and microbial tests on rice kernels, covering both raw materials and final products. This comprehensive approach ensures that the factory adheres to rigorous cleanliness and safety standards, contributing to the production of high-quality FRKs.
Equipment
The equipment, constructed from stainless steel, plastic, and other food-grade materials, features an easily maintainable design. Regular semi-annual preventive maintenance guarantees an efficient, crack-free, rust-free, and dent-free operational system.
Personal hygiene
Adherence to good manufacturing practices ensures the establishment of robust personal hygiene standards among employees, as per the Standard Operating Procedure (SOP). Employees strictly comply with personal hygiene instructions, donning uniforms, gloves, and hairnets to prevent any potential cross-contamination. The guidelines explicitly prohibit actions such as placing fingers in sensitive areas like the mouth, ears, eyes, and nose. Additionally, activities like eating, spitting, chewing, drinking, and smoking are strictly forbidden during food equipment and materials handling and operations. A committed quality control supervisor ensures these practices are followed and keeps records verifying that employees wash and sanitize their hands before starting work and after returning from the washrooms.
Water supply
The factory features a dedicated underground water supply and storage system, delivering sufficient clean, and drinking water post reverse osmosis treatment. This portable, treated water undergoes hourly testing and adheres to Bangladesh’s national water quality standards.
Storage and transportation
Every storage room, except the maintenance store, is meticulously cleaned, sanitized, and maintained with controlled temperature and humidity, featuring air conditioning. The maintenance store is monitored and verified using a hygrometer and data logger. Regular inspections ensure a consistent environment, mitigating hazards, and facilitating the production of finished products, which maintain the same quality from the production to the consumer end. The factory is equipped with proper transportation facilities, overseeing temperature, and separation through the transit of both types of materials, such as raw and finished goods, to ensure compliance with food safety standards.
Sanitation program
Maintaining a sanitary and culturally approachable atmosphere is crucial for storing any kind of goods to ensure providing a safe product for the consumer.
Pest control management
Pest control in the factory aims to remove various kinds and sizes of pests, including human-made incidents. Modern pest management programs, surpassing traditional insecticide spraying and trapping techniques, are employed monthly by factory employees and contracted third-party service providers.
Waste management
The company operates its waste treatment factory with a comprehensive drainage, storage, and disposal system. It consistently checks and tests incoming waste and discharging wastewater to ensure compliance with international standards.
Traceability
Accurate record-keeping includes lot identification numbers, batch numbers, and key dates such as the arrival of raw materials and vitamin premix, production, and premix making. Strict adherence to the First In, First Out (FIFO) system is maintained in the store warehouse for both raw and packaging materials.
Training
Quality control officers conduct bi-weekly training sessions for all employees, addressing essential aspects such as maintaining personal and operational hygiene, operational and handling safety, organizational law, policy and regulations, production processes, and food safety and distribution issues.
Production process
The incoming white, parboiled, or broken rice undergoes in-house crushing using a grinder. The resulting powder is weighed and blended with vitamin premix such as palmitate or acetate as Vitamin A, thiamine mononitrate as vitamin B1, cyanocobalamin as B12, folic acid as B9, ferric pyrophosphate as Fe, and zinc oxide as Zn. The dry mixture undergoes preconditioning in a preconditioned at 70 to 80 °C, then moves to a screw conveyor. A hot extruder, maintained at 85 to 95 °C for 30 sec, produces FRKs. The FRK is subsequently transferred to a roller drier to eliminate excess moisture, followed by a multi-layer dryer at 70 to 90 °C. After cooling and metal detection, the FRK is conveyed to the packaging room, utilizing a food-grade three-layer craft paper. The process flow is illustrated in Figure 1.

Figure 1: Process flow diagram of Fortified Rice Kernels (FRKs)
Hazards identification
A hazard, as defined by Xi et al. (2023) and Yaashikaa et al. (2023), is a property capable of rendering a finished product unsafe for human consumption. Hazards are categorized into physical, chemical, and biological hazards. The analysis and documentation of hazards is an ongoing process, beginning with identifying potential threats posed by raw materials to human health. Tables 2 and 3 define the three types of risks associated with all additives and raw materials and the processing line for rice fortification production. From Table 3, the processed FRKs are stored at the storage area, which can be contaminated with bacteria and fungi, resulting in chemical and biological hazards. We can remove them using different management tools such as total quality management, good manufacturing practices, and modified storage conditions. The grinding and mixing of the broken rice should be performed by quality personnel. The operations' time and temperature are critical for the FRK, which can be maintained for microbial growth. The metal detection system is utilized to identify the presence of metals.
A hazard, as defined by Xi et al. (2023) and Yaashikaa et al. (2023), is a property capable of rendering a finished product unsafe for human consumption. Hazards are categorized into physical, chemical, and biological hazards. The analysis and documentation of hazards is an ongoing process, beginning with identifying potential threats posed by raw materials to human health. Tables 2 and 3 define the three types of risks associated with all additives and raw materials and the processing line for rice fortification production. From Table 3, the processed FRKs are stored at the storage area, which can be contaminated with bacteria and fungi, resulting in chemical and biological hazards. We can remove them using different management tools such as total quality management, good manufacturing practices, and modified storage conditions. The grinding and mixing of the broken rice should be performed by quality personnel. The operations' time and temperature are critical for the FRK, which can be maintained for microbial growth. The metal detection system is utilized to identify the presence of metals.
Table 2: Hazards in operations, ingredients, and raw material investigation chart
| Ingredients and Materials | Hazards | Preventive Measure | |
| Rice flour source (White rice or parboiled rice, or broken rice) | Biological hazard | Pathogens | Sanitize apparatus and instruments properly, ensure individual hygiene, and material handling |
| Chemical hazard | Adulterants, allergens | ||
| Physical hazard | Foreign particles | ||
| Vitamin premix | Biological hazard | Pathogens | Maintain quality assurance, Maintain first-in-first-out Ensure operational hygiene |
| Chemical hazard | Adulterants, allergens | ||
| Physical hazard | Foreign particles | ||
| Packaging Materials | Chemical hazard | Ink | Food grade ink and other material utilization, The quality assurance personnel ensure quality Strictly maintain store temperature below 20 ºC |
| Physical hazard | Presence of the foreign particles | ||
Table 3: Hazards analysis chart for Fortified Rice Kernels (FRKs) processing step
| Process step | Hazards | Preventive measure | |
| Raw Material Storage (Rice flour source and vitamin premix) | Biological hazard | Escherichia coli, Salmonella spp. | Instruments and apparatus should be accurately set, equipment cleaned and sanitized before operation, and confirmed individual hygiene and materials handling |
| Chemical hazard | Allergen, antibiotic residue | ||
| Mixing | Biological hazard | Pathogen from the grinder | All equipment is clean and dry, Ensured operational hygiene. |
| Chemical hazard | Sanitizer, cleaning agent |
||
| Physical hazard | Foreign materials | ||
| Drying | Biological hazard | Microorganisms (pathogens), if not killed | The drying temperature of the FRK is above 100 ºC, The drying time of the FRK is 30 sec |
| Cooling | No hazard is associated with this step | Air cooling utilization, Pieces of equipment should be cleaned and sanitized before use |
|
| Metal detection | Physical hazard | Any foreign particle can enter | Detect metals: Stainless Steel (1.2 mm) Ferrous (1.25 mm) Non-ferrous (1.2 mm) |
| Packing the fortified rice kernels | Physical hazard | Any foreign particle can enter | Packing machine settings should be properly set, and the provisional container should be sanitized. Calibrate all kinds of equipment, including the weighing scales. Confirmed individual hygiene and materials handling |
| Storage in the separate room and distributions | Biological hazard | Growth of microorganisms | Hygienic and well-maintained storage facilities. Distribution: clean before utilizing the delivery truck |
CCP determination
CCPs are the pivotal stage where a specific control measure must be operational to eradicate any kind of hazard, including food and health and minimized to a tolerable level (De Oliveira et al., 2016; Mosso et al., 2025; Motarjemi and Warren, 2023). In this process, the drying temperature is designated as a CCP, and its core temperature validates the efficacy of the drying process (Asaduzzaman, 2021; De Oliveira et al., 2016; Mosso et al., 2025). Figure 1 provides a decision tree employed to categorize CCPs for raw ingredients, as detailed in Table 2. The corresponding decision tree in Table 2 is applied to the identify CCPs for operational steps, as is also done in Table 4. Again, utilizing the decision tree (Figure 1), we can determine the CCPs for the different processing steps in Table 5, where we can see that drying by the oven can cause hazards due to the temperature and time variation, which can cause biological hazards.
CCPs are the pivotal stage where a specific control measure must be operational to eradicate any kind of hazard, including food and health and minimized to a tolerable level (De Oliveira et al., 2016; Mosso et al., 2025; Motarjemi and Warren, 2023). In this process, the drying temperature is designated as a CCP, and its core temperature validates the efficacy of the drying process (Asaduzzaman, 2021; De Oliveira et al., 2016; Mosso et al., 2025). Figure 1 provides a decision tree employed to categorize CCPs for raw ingredients, as detailed in Table 2. The corresponding decision tree in Table 2 is applied to the identify CCPs for operational steps, as is also done in Table 4. Again, utilizing the decision tree (Figure 1), we can determine the CCPs for the different processing steps in Table 5, where we can see that drying by the oven can cause hazards due to the temperature and time variation, which can cause biological hazards.
Table 4: Rice fortification ingredients decision matrix
Table 5: Analysis of hazards in rice fortification process steps and Critical Control Point (CCP) decision matrix
| Guidelines for identifying Critical Control Points (CCPs) Step 1: Determine whether the raw materials present any potential hazards. –If no, then the step is not a CCP –If yes, move to Step 2 Step 2: Will the identified hazards be eliminated or reduced through further processing? –If no, the step is a CCP –If yes, proceed to Step 3 Step 3: Is there a risk of cross-contamination to the facility or other products that cannot be adequately controlled? –If no, it is not a CCP –If yes, the step is considered a CCP |
||||||
| Raw material |
Kinds of Hazard |
Question 1 |
Question 2 |
Question 3 |
CCP | Remarks |
| Rice flour source (White rice or parboiled rice, or broken rice) | ||||||
| Biological hazard | Pathogen | Yes | Yes | No | No | Drying removes the microbial hazards |
| Chemical hazard | Adulterants, allergens | Yes | Yes | No | No | The received product can be critical |
| Physical hazard | Foreign Materials | No | - | - | No | Proper physical inspection, personal hygiene, and Good Manufacturing Practice (GMP) |
| Vitamin premix | ||||||
| Biological hazard | Pathogen | Yes | Y | No | No | Drying process, personal hygiene, GMPs, and food storage |
| Chemical hazard | Adulterants | Yes | No | - | Yes | Critical raw product may be received |
| Physical hazard | Foreign materials | Yes | No | - | Yes | Critical raw product may be received |
| Packaging Materials | ||||||
| Chemical hazard | Adulterants | Yes | No | - | Yes | Critical raw product may be received |
| Physical hazard | Foreign materials | Yes | No | - | Yes | Critical raw product may be received |
Table 5: Analysis of hazards in rice fortification process steps and Critical Control Point (CCP) decision matrix
| Guidelines for determining CCPs Step 1: Does this step involve a hazard with sufficient risk and severity to require control? –If no, then this step is not a CCP –If yes, move to Step 2 Step 2: Could any preventive actions, themselves, at this step introduce a hazard? –If yes, continue to Step 3 –If no, proceed to Step 2a Step 2a: Are specific safety controls needed at this step? –If yes, adjust the process, step, or product accordingly –If no, this step is not a CCP Step 3: Is it essential to control the hazard at this step to ensure consumer safety? –If yes, this is identified as a CCP –If no, then it is not a CCP |
||||||
| Process step | Kinds of hazards | Question 1 |
Question 2 |
Question 3 |
CCP | Remarks |
| Raw Material Storage (Rice flour source and vitamin premix) | ||||||
| Biological hazard | Pathogen | Yes | Yes | Yes | Yes | Storage time, temperature, and operational hygiene are critical |
| Chemical hazard | Antibiotics, Sanitizer, and Cleaner |
No | No | Several prerequisite programs, such as water quality and sanitation systems, are applicable. | ||
| Physical hazard | Foreign particles | Yes | Yes | No | No | Several prerequisite programs, such as operational and personal hygiene and pest control management, are applicable. |
| Mixing | ||||||
| Biological hazard | Pathogen | Yes | Yes | No | No | Personal hygiene, pest control, maintenance and cleaning |
| Chemical hazard | Sanitizer and cleaner |
No | No | Several prerequisite programs, such as operational and preventive maintenance and Cleaning in Place (CIP), and sanitation system. | ||
| Physical hazard | Foreign particles | Yes | Yes | No | No | Several prerequisite programs, such as pest control management, operational, and personal hygiene |
| Drying | ||||||
| Biological hazard | Pathogen | Yes | Yes | Yes | Yes | Correct drying temperature and time are critical, The prerequisite program will confirm treated water quality |
| Metal detection | ||||||
| Physical hazard | Any foreign particle enters | Yes | Yes | Yes | Yes | Foreign materials (ferrous, Non-ferrous, and stainless steel) are critical. |
| Packing | ||||||
| Biological hazard | Microorganisms/ Pathogen | No | No | Prerequisite program: individual hygiene and hygiene system | ||
| Physical hazard | Foreign particles | Yes | Yes | Yes | Yes | The presence of foreign materials must be critical |
| Storage and distribution | ||||||
| Biological hazard | Pathogen | Yes | Yes | Yes | Yes | Storage and distribution time and temperature must be strictly maintained below 20 ºC and must be critical |
The meticulous consideration of vitamin premix and packaging materials is imperative due to the heat-sensitive chemical particles and components present in these products, which are commonly susceptible to adulteration in Bangladesh. Both chemical and physical hazards associated with packaging materials and vitamin premixes are deemed critical. The precise control of time and temperature during drying, with a focus on core temperature, is a crucial factor for FRKs to ensure the destruction of pathogens. The establishment of a separate packaging room proves highly beneficial in maintaining product color and preventing microbial growth. Furthermore, the incorporation of a metal detection system is instrumental in eliminating the presence of foreign and metal materials.
A similar result was found in the study by Matéyendou and Ouézou Yaovi (2013), where they implemented the FSMS to small and medium food businesses in Togo, where they intend to export products to the international market. They identified several hazards due to the employees (Matéyendou and Ouézou Yaovi, 2013). According to the study, the West African Economic and Monetary :union: (WAEMU) suggest that FSMS is mandatory to improve the facilities environment and produce safe food for the consumers and the international market.
According to Kirezieva et al. (2013), the most significant hazard is the presence of foreign materials in the final product, which can impact the brand value of the company and create health and trust issues regarding the factory's products.
FSMS control chart
Table 6 illustrates a comprehensive overview of potential critical hazards, encompassing physical, chemical, and microbial risks that may arise throughout the manufacturing processes in the rice fortification factory, denoted as the FSMS control chart. This chart provides a comprehensive overview by identifying not only the CCPs but also detailing the associated control measures, specific critical limits, monitoring procedures with their respective frequencies, and both preventive, and corrective actions for each critical point. It also includes the relevant documentation and assigns responsibility to designated personnel. The highlighted control points correspond to potential hazards found in both raw materials and production stages.
Building on findings from previous research, the FSMS control chart integrates detailed hazard descriptions related to the product, critical limits for each CCP, observation protocols, assigned personnel for each control point, monitoring methods, and corrective actions (Asaduzzaman, 2021; Mosso et al., 2025). Highlighting the significance of consistent monitoring and thorough documentation, the Codex Alimentarius commission, as cited by Asaduzzaman (2021), stresses the inclusion of these elements in the FSMS plan for FRKs. In this fortification facility, a total of two CCPs and two control points have been identified.
The tool is now being used in combination with a similarly designed instrument created to audit FSMS activities, with the aim to establish the status of food safety management systems both at the primary production level and across entire supply chains, both within Europe and internationally. Results of such quantitative surveys are expected to be reported in the near future. Future research will integrate microbiological data with system-level investigation to facilitate risk profiling by specific supply chain phases, product categories, or nations. The findings will inform the creation of targeted quality assurance requirements, guidelines, and recommendations for fresh produce supply chains, customized according to climate zones, national contexts, supply chain actors, and product categories.
A similar result was found in the study by Matéyendou and Ouézou Yaovi (2013), where they implemented the FSMS to small and medium food businesses in Togo, where they intend to export products to the international market. They identified several hazards due to the employees (Matéyendou and Ouézou Yaovi, 2013). According to the study, the West African Economic and Monetary :union: (WAEMU) suggest that FSMS is mandatory to improve the facilities environment and produce safe food for the consumers and the international market.
According to Kirezieva et al. (2013), the most significant hazard is the presence of foreign materials in the final product, which can impact the brand value of the company and create health and trust issues regarding the factory's products.
FSMS control chart
Table 6 illustrates a comprehensive overview of potential critical hazards, encompassing physical, chemical, and microbial risks that may arise throughout the manufacturing processes in the rice fortification factory, denoted as the FSMS control chart. This chart provides a comprehensive overview by identifying not only the CCPs but also detailing the associated control measures, specific critical limits, monitoring procedures with their respective frequencies, and both preventive, and corrective actions for each critical point. It also includes the relevant documentation and assigns responsibility to designated personnel. The highlighted control points correspond to potential hazards found in both raw materials and production stages.
Building on findings from previous research, the FSMS control chart integrates detailed hazard descriptions related to the product, critical limits for each CCP, observation protocols, assigned personnel for each control point, monitoring methods, and corrective actions (Asaduzzaman, 2021; Mosso et al., 2025). Highlighting the significance of consistent monitoring and thorough documentation, the Codex Alimentarius commission, as cited by Asaduzzaman (2021), stresses the inclusion of these elements in the FSMS plan for FRKs. In this fortification facility, a total of two CCPs and two control points have been identified.
The tool is now being used in combination with a similarly designed instrument created to audit FSMS activities, with the aim to establish the status of food safety management systems both at the primary production level and across entire supply chains, both within Europe and internationally. Results of such quantitative surveys are expected to be reported in the near future. Future research will integrate microbiological data with system-level investigation to facilitate risk profiling by specific supply chain phases, product categories, or nations. The findings will inform the creation of targeted quality assurance requirements, guidelines, and recommendations for fresh produce supply chains, customized according to climate zones, national contexts, supply chain actors, and product categories.
Table 6: Food Safety Management System (FSMS) control chart for rice fortification production
| Process Step and CCP No |
Hazards | Critical limits |
Monitoring procedure |
Frequency | Preventive measure |
Corrective action |
Record | Responsible person |
| Raw ingredients and packaging material; CP#1 |
Microbial Chemical and Physical Contaminations |
Pure product to be used |
Apply supply quality control or assurance |
Each supply from the supplier | Test received raw material; Check MSDS; Review and approve supplier list; Specification of the material |
Change supplier or brand in interval; Employee internal and external training for raw material testing |
Materials receiving and testing report |
Assigned receiver (QC/QA) |
| Drying; CCP#1 |
Survival of pathogens |
Drying temperature 90 °C | Check the drying time and core temperature. Follow up the time and temperature and record keeping |
Each batch | Check the temperature of the product |
Adjust the temperature and time by setting the equipment; Call the Maintenance team to repair |
Time and core Temperature of the product log. Maintenance register |
Assigned executive (QC/QA) |
| Metal detection; CCP#2 |
Foreign materials present | Detect metals: stainless still (1.2 mm) ferrous (1.25 mm) non-ferrous (1.2 mm) |
Record keeping |
Each batch | Check foreign materials | Calibrate the metal detector; Call the maintenance team to repair |
Time and corrective action of product log; Maintenance register |
Assigned executive (QC/QA) |
| Packing; CP#2 |
Physical contamination |
No foreign material; No leakage |
Metal detection system; Visual Inspection by packing supervisor |
Each pack | Personal hygiene and physical inspection |
Retain, rework, or discard based on identification of foreign material |
Metal detection system report, inspection report |
Assigned executive (QC/QA) |
Conclusions
To attain and enhance product safety and quality, this study contributes to developing a FSMS model specifically tailored for FRKs. The principles and objectives of FSMS were applied to formulate the rice fortification model. Simplifying the FSMS plan and prerequisite programs, including considerations for location, equipment, water supply, etc., were intended to address potential hazards before production. This study utilized product descriptions to alert purchasers and suppliers to potential hazards such as physical, chemical, and biological risks in the finished products. Subsequently, potential hazard control points were identified in raw materials and the production process, accompanied by corresponding preventive measures. CCPs were identified and delineated through decision trees that guide responses to key questions. Finally, a comprehensive FSMS control chart was crafted, incorporating key components of FSMS principles. This study identified two CCPs within the rice fortification factory's manufacturing process: the supply of raw materials and primary packaging materials, and the adherence to proper time, and temperature during drying. Additionally, adequate observation and inspection during packing were emphasized to mitigate the presence of foreign and metal materials in the final product.
Author contributions
T.K. and M.A. designed the study, conducted the experiments, analyzed data, and wrote the manuscript; M.A. reviewed the manuscript, conducted validation, supervised the study, acquired funding, and administered the project. Both authors read and approved the final manuscript.
Acknowledgments
This research received an administrative grant from the State University of Bangladesh.
Conflicts of interest
The authors declare that there is no conflict of interest.
Funding
The State University of Bangladesh provided M. Asaduzzaman's funding for industry process and environment improvement. M. Asaduzzaman reports a relationship with the State University of Bangladesh that includes employment.
Ethical consideration
Not applicable.
References
Asaduzzaman M. (2021). The Implementation of hazard analysis critical control point (HACCP) Plan for Chicken Nugget Plant. Asian Food Science Journal. 20: 11-24. [DOI: 10.9734/ AFSJ/2021/v20i530295]
Bangladesh Standards and Testing Institution (BSTI). (2015). Bangladesh standard: specification for fortified rice. national standard No. BDS 1897:2015. URL: https:// bsti.portal.gov.bd/ sites/default/files/files/bsti.portal.gov.bd/notices/e75417bf_2fee_4667_89b3_0895dd63f2c1/2022-04-03-06-19- 2c2438e4e09a6e387d672a1c6b123dae.pdf. Accessed 01 June 2025.
De Boeck E., Jacxsens L., Bollaerts M., Uyttendaele M., Vlerick P. (2016). Interplay between food safety climate, food safety management system and microbiological hygiene in farm butcheries and affiliated butcher shops. Food Control. 65: 78-91. [DOI: 10.1016/j.foodcont.2016.01.014]
De Oliveira C.A.F., Da Cruz A.G., Tavolaro P., Corassin C.H. (2016). Food safety: good manufacturing practices (GMP), sanitation standard operating procedures (SSOP), hazard analysis and critical control point (HACCP). In: Barros-Velázquez J. (Editor.). Antimicrobial food packaging. 2nd edition. Academic Press, United States. pp: 129-139. [DOI: 10.1016/B978-0-12-800723-5.00010-3]
De Lima A.B.S., Becerra C.E.T., Feitosa A.D., De Albuquerque A.P.G., De Melo F.J.C., De Medeiros D.D. (2025). Effective practices for implementing quality control circles aligned with iso quality standards: insights from employees and managers in the food industry. Standards. 5: 6. [DOI: 10.3390/standards5010006]
De Oleo D.D., Manning L., McIntyre L., Randall N., Nayak R. (2024). The application of systematic accident analysis tools to investigate food safety incidents. Comprehensive Reviews in Food Science and Food Safety. 23: e13344. [DOI: 10.1111/1541-4337.13344]
Dzwolak W. (2019). Assessment of HACCP plans in standardized food safety management systems – the case of small-sized Polish food businesses. Food Control. 106: 106716. [DOI: 10.1016/j.foodcont.2019.106716]
Kirezieva K., Nanyunja J., Jacxsens L., Van Der Vorst J.G.A.J., Uyttendaele M., Luning, P.A. (2013). Context factors affecting design and operation of food safety management systems in the fresh produce chain. Trends in Food Science and Technology. 32: 108-127. [DOI: 10.1016/j.tifs.2013.06.001]
Matéyendou L., Ouézou Yaovi A. (2013). Integration of food safety management systems in the design of small and medium food businesses in Togo. Environnement Risques and Santé. 12: 521-529. [DOI: 10.1684/ers.2013.0658]. [French with English abstract]
Mosso J., Reyes G.A., Kowalcyk B., Davis D.A. (2025). Testing program critical control points (TP-CCP): characterizing and optimizing decision-making power in food safety testing. Journal of Food Protection. 88: 100528. [DOI: 10.1016/j.jfp.2025.100528]
Motarjemi Y., Warren B.R. (2023). Hazard analysis and critical control point system (HACCP). In: Andersen V., Lelieveld H., Motarjemi Y. (Editors.). Food Safety Management. 2nd edition. Academic Press, United States. pp: 799-818. [DOI: 10.1016/B978-0-12-820013-1.00017-6]
Nandeep E.R., Mahajan H., Mummadi M.K., Sairam C., Venkatesh K., Kadiyam J., Meshram I., Pagidoju S., Reddy V.R., Panda H., Pullakandham R., Geddam, J.J.B., et al. (2024). Implementation, delivery, and utilization of iron fortified rice supplied through public distribution system across different states in India: an exploratory mixed-method study. Plos Global Public Healt. 4: e0003533. [DOI: 10.1371/ journal.pgph.0003533]
Okpala C.O.R., Korzeniowska M. (2023). Understanding the relevance of quality management in agro-food product industry: from ethical considerations to assuring food hygiene quality safety standards and its associated processes. Food Reviews International. 39: 1879-1952. [DOI: 10.1080/ 87559129.2021.1938600]
Oruc A., Chowdhury N., Gkioulos V. (2024). A modular cyber security training programme for the maritime domain. International Journal of Information Security. 23: 1477-1512. [DOI: 10.1007/s10207-023-00799-4]
Rincon-Ballesteros L., Lannelongue G., González-Benito J. (2024). Cross-continental insights: comparing food safety management systems in Europe and Latin America. Food Control. 164: 110552. [DOI: 10.1016/j.foodcont.2024.110552]
Thakur C., Kaushal M., Vaidya D., Verma A.K., Gupta A., Sharma R. (2025). Unlocking the potential of spray drying for agro-products: exploring advanced techniques, carrier agents, applications, and limitations. Food and Bioprocess Technology. 18: 1181-1220. [DOI: 10.1007/s11947-024-03544-4]
Xi D., Lin N., Gori A. (2023). Increasing sequential tropical cyclone hazards along the US East and Gulf coasts. Nature Climate Change. 13: 258-265. [DOI: 10.1038/s41558-023-01595-7]
Yaashikaa P.R., Kamalesh R., Senthil Kumar P., Saravanan A., Vijayasri K., Rangasamy G. (2023). Recent advances in
edible coatings and their application in food packaging.
Food Research International. 173: 113366. [DOI: 10.1016/ j.foodres.2023.113366]
To attain and enhance product safety and quality, this study contributes to developing a FSMS model specifically tailored for FRKs. The principles and objectives of FSMS were applied to formulate the rice fortification model. Simplifying the FSMS plan and prerequisite programs, including considerations for location, equipment, water supply, etc., were intended to address potential hazards before production. This study utilized product descriptions to alert purchasers and suppliers to potential hazards such as physical, chemical, and biological risks in the finished products. Subsequently, potential hazard control points were identified in raw materials and the production process, accompanied by corresponding preventive measures. CCPs were identified and delineated through decision trees that guide responses to key questions. Finally, a comprehensive FSMS control chart was crafted, incorporating key components of FSMS principles. This study identified two CCPs within the rice fortification factory's manufacturing process: the supply of raw materials and primary packaging materials, and the adherence to proper time, and temperature during drying. Additionally, adequate observation and inspection during packing were emphasized to mitigate the presence of foreign and metal materials in the final product.
Author contributions
T.K. and M.A. designed the study, conducted the experiments, analyzed data, and wrote the manuscript; M.A. reviewed the manuscript, conducted validation, supervised the study, acquired funding, and administered the project. Both authors read and approved the final manuscript.
Acknowledgments
This research received an administrative grant from the State University of Bangladesh.
Conflicts of interest
The authors declare that there is no conflict of interest.
Funding
The State University of Bangladesh provided M. Asaduzzaman's funding for industry process and environment improvement. M. Asaduzzaman reports a relationship with the State University of Bangladesh that includes employment.
Ethical consideration
Not applicable.
References
Asaduzzaman M. (2021). The Implementation of hazard analysis critical control point (HACCP) Plan for Chicken Nugget Plant. Asian Food Science Journal. 20: 11-24. [DOI: 10.9734/ AFSJ/2021/v20i530295]
Bangladesh Standards and Testing Institution (BSTI). (2015). Bangladesh standard: specification for fortified rice. national standard No. BDS 1897:2015. URL: https:// bsti.portal.gov.bd/ sites/default/files/files/bsti.portal.gov.bd/notices/e75417bf_2fee_4667_89b3_0895dd63f2c1/2022-04-03-06-19- 2c2438e4e09a6e387d672a1c6b123dae.pdf. Accessed 01 June 2025.
De Boeck E., Jacxsens L., Bollaerts M., Uyttendaele M., Vlerick P. (2016). Interplay between food safety climate, food safety management system and microbiological hygiene in farm butcheries and affiliated butcher shops. Food Control. 65: 78-91. [DOI: 10.1016/j.foodcont.2016.01.014]
De Oliveira C.A.F., Da Cruz A.G., Tavolaro P., Corassin C.H. (2016). Food safety: good manufacturing practices (GMP), sanitation standard operating procedures (SSOP), hazard analysis and critical control point (HACCP). In: Barros-Velázquez J. (Editor.). Antimicrobial food packaging. 2nd edition. Academic Press, United States. pp: 129-139. [DOI: 10.1016/B978-0-12-800723-5.00010-3]
De Lima A.B.S., Becerra C.E.T., Feitosa A.D., De Albuquerque A.P.G., De Melo F.J.C., De Medeiros D.D. (2025). Effective practices for implementing quality control circles aligned with iso quality standards: insights from employees and managers in the food industry. Standards. 5: 6. [DOI: 10.3390/standards5010006]
De Oleo D.D., Manning L., McIntyre L., Randall N., Nayak R. (2024). The application of systematic accident analysis tools to investigate food safety incidents. Comprehensive Reviews in Food Science and Food Safety. 23: e13344. [DOI: 10.1111/1541-4337.13344]
Dzwolak W. (2019). Assessment of HACCP plans in standardized food safety management systems – the case of small-sized Polish food businesses. Food Control. 106: 106716. [DOI: 10.1016/j.foodcont.2019.106716]
Kirezieva K., Nanyunja J., Jacxsens L., Van Der Vorst J.G.A.J., Uyttendaele M., Luning, P.A. (2013). Context factors affecting design and operation of food safety management systems in the fresh produce chain. Trends in Food Science and Technology. 32: 108-127. [DOI: 10.1016/j.tifs.2013.06.001]
Matéyendou L., Ouézou Yaovi A. (2013). Integration of food safety management systems in the design of small and medium food businesses in Togo. Environnement Risques and Santé. 12: 521-529. [DOI: 10.1684/ers.2013.0658]. [French with English abstract]
Mosso J., Reyes G.A., Kowalcyk B., Davis D.A. (2025). Testing program critical control points (TP-CCP): characterizing and optimizing decision-making power in food safety testing. Journal of Food Protection. 88: 100528. [DOI: 10.1016/j.jfp.2025.100528]
Motarjemi Y., Warren B.R. (2023). Hazard analysis and critical control point system (HACCP). In: Andersen V., Lelieveld H., Motarjemi Y. (Editors.). Food Safety Management. 2nd edition. Academic Press, United States. pp: 799-818. [DOI: 10.1016/B978-0-12-820013-1.00017-6]
Nandeep E.R., Mahajan H., Mummadi M.K., Sairam C., Venkatesh K., Kadiyam J., Meshram I., Pagidoju S., Reddy V.R., Panda H., Pullakandham R., Geddam, J.J.B., et al. (2024). Implementation, delivery, and utilization of iron fortified rice supplied through public distribution system across different states in India: an exploratory mixed-method study. Plos Global Public Healt. 4: e0003533. [DOI: 10.1371/ journal.pgph.0003533]
Okpala C.O.R., Korzeniowska M. (2023). Understanding the relevance of quality management in agro-food product industry: from ethical considerations to assuring food hygiene quality safety standards and its associated processes. Food Reviews International. 39: 1879-1952. [DOI: 10.1080/ 87559129.2021.1938600]
Oruc A., Chowdhury N., Gkioulos V. (2024). A modular cyber security training programme for the maritime domain. International Journal of Information Security. 23: 1477-1512. [DOI: 10.1007/s10207-023-00799-4]
Rincon-Ballesteros L., Lannelongue G., González-Benito J. (2024). Cross-continental insights: comparing food safety management systems in Europe and Latin America. Food Control. 164: 110552. [DOI: 10.1016/j.foodcont.2024.110552]
Thakur C., Kaushal M., Vaidya D., Verma A.K., Gupta A., Sharma R. (2025). Unlocking the potential of spray drying for agro-products: exploring advanced techniques, carrier agents, applications, and limitations. Food and Bioprocess Technology. 18: 1181-1220. [DOI: 10.1007/s11947-024-03544-4]
Xi D., Lin N., Gori A. (2023). Increasing sequential tropical cyclone hazards along the US East and Gulf coasts. Nature Climate Change. 13: 258-265. [DOI: 10.1038/s41558-023-01595-7]
Yaashikaa P.R., Kamalesh R., Senthil Kumar P., Saravanan A., Vijayasri K., Rangasamy G. (2023). Recent advances in
edible coatings and their application in food packaging.
Food Research International. 173: 113366. [DOI: 10.1016/ j.foodres.2023.113366]
[*] Corresponding author (M. Asaduzzaman)
* E-mail: bitatipu@gmail.com
ORCID ID: https://orcid.org/0000-0001-7202-961X
* E-mail: bitatipu@gmail.com
ORCID ID: https://orcid.org/0000-0001-7202-961X
Type of Study: Original article |
Subject:
Special
Received: 24/12/11 | Accepted: 25/06/18 | Published: 25/06/22
Received: 24/12/11 | Accepted: 25/06/18 | Published: 25/06/22
References
1. Asaduzzaman M. (2021). The Implementation of hazard analysis critical control point (HACCP) Plan for Chicken Nugget Plant. Asian Food Science Journal. 20: 11-24. [DOI: 10.9734/ AFSJ/2021/v20i530295] [DOI:10.9734/afsj/2021/v20i530295]
2. Bangladesh Standards and Testing Institution (BSTI). (2015). Bangladesh standard: specification for fortified rice. national standard No. BDS 1897:2015. URL: https:// bsti.portal.gov.bd/ sites/default/files/files/bsti.portal.gov.bd/notices/e75417bf_2fee_4667_89b3_0895dd63f2c1/2022-04-03-06-19- 2c2438e4e09a6e387d672a1c6b123dae.pdf. Accessed 01 June 2025.
3. De Boeck E., Jacxsens L., Bollaerts M., Uyttendaele M., Vlerick P. (2016). Interplay between food safety climate, food safety management system and microbiological hygiene in farm butcheries and affiliated butcher shops. Food Control. 65: 78-91. [DOI: 10.1016/j.foodcont.2016.01.014] [DOI:10.1016/j.foodcont.2016.01.014]
4. De Oliveira C.A.F., Da Cruz A.G., Tavolaro P., Corassin C.H. (2016). Food safety: good manufacturing practices (GMP), sanitation standard operating procedures (SSOP), hazard analysis and critical control point (HACCP). In: Barros-Velázquez J. (Editor.). Antimicrobial food packaging. 2nd edition. Academic Press, United States. pp: 129-139. [DOI: 10.1016/B978-0-12-800723-5.00010-3] [DOI:10.1016/B978-0-12-800723-5.00010-3]
5. De Lima A.B.S., Becerra C.E.T., Feitosa A.D., De Albuquerque A.P.G., De Melo F.J.C., De Medeiros D.D. (2025). Effective practices for implementing quality control circles aligned with iso quality standards: insights from employees and managers in the food industry. Standards. 5: 6. [DOI: 10.3390/standards5010006] [DOI:10.3390/standards5010006]
6. De Oleo D.D., Manning L., McIntyre L., Randall N., Nayak R. (2024). The application of systematic accident analysis tools to investigate food safety incidents. Comprehensive Reviews in Food Science and Food Safety. 23: e13344. [DOI: 10.1111/1541-4337.13344] [DOI:10.1111/1541-4337.13344] [PMID]
7. Dzwolak W. (2019). Assessment of HACCP plans in standardized food safety management systems - the case of small-sized Polish food businesses. Food Control. 106: 106716. [DOI: 10.1016/j.foodcont.2019.106716] [DOI:10.1016/j.foodcont.2019.106716]
8. Kirezieva K., Nanyunja J., Jacxsens L., Van Der Vorst J.G.A.J., Uyttendaele M., Luning, P.A. (2013). Context factors affecting design and operation of food safety management systems in the fresh produce chain. Trends in Food Science and Technology. 32: 108-127. [DOI: 10.1016/j.tifs.2013.06.001] [DOI:10.1016/j.tifs.2013.06.001]
9. Matéyendou L., Ouézou Yaovi A. (2013). Integration of food safety management systems in the design of small and medium food businesses in Togo. Environnement Risques and Santé. 12: 521-529. [DOI: 10.1684/ers.2013.0658]. [French with English abstract]
10. Mosso J., Reyes G.A., Kowalcyk B., Davis D.A. (2025). Testing program critical control points (TP-CCP): characterizing and optimizing decision-making power in food safety testing. Journal of Food Protection. 88: 100528. [DOI: 10.1016/j.jfp.2025.100528] [DOI:10.1016/j.jfp.2025.100528] [PMID]
11. Motarjemi Y., Warren B.R. (2023). Hazard analysis and critical control point system (HACCP). In: Andersen V., Lelieveld H., Motarjemi Y. (Editors.). Food Safety Management. 2nd edition. Academic Press, United States. pp: 799-818. [DOI: 10.1016/B978-0-12-820013-1.00017-6] [DOI:10.1016/B978-0-12-820013-1.00017-6]
12. Nandeep E.R., Mahajan H., Mummadi M.K., Sairam C., Venkatesh K., Kadiyam J., Meshram I., Pagidoju S., Reddy V.R., Panda H., Pullakandham R., Geddam, J.J.B., et al. (2024). Implementation, delivery, and utilization of iron fortified rice supplied through public distribution system across different states in India: an exploratory mixed-method study. Plos Global Public Healt. 4: e0003533. [DOI: 10.1371/ journal.pgph.0003533] [DOI:10.1371/journal.pgph.0003533] [PMID] [PMCID]
13. Okpala C.O.R., Korzeniowska M. (2023). Understanding the relevance of quality management in agro-food product industry: from ethical considerations to assuring food hygiene quality safety standards and its associated processes. Food Reviews International. 39: 1879-1952. [DOI: 10.1080/ 87559129.2021.1938600] [DOI:10.1080/87559129.2021.1938600]
14. Oruc A., Chowdhury N., Gkioulos V. (2024). A modular cyber security training programme for the maritime domain. International Journal of Information Security. 23: 1477-1512. [DOI: 10.1007/s10207-023-00799-4] [DOI:10.1007/s10207-023-00799-4]
15. Rincon-Ballesteros L., Lannelongue G., González-Benito J. (2024). Cross-continental insights: comparing food safety management systems in Europe and Latin America. Food Control. 164: 110552. [DOI: 10.1016/j.foodcont.2024.110552] [DOI:10.1016/j.foodcont.2024.110552]
16. Thakur C., Kaushal M., Vaidya D., Verma A.K., Gupta A., Sharma R. (2025). Unlocking the potential of spray drying for agro-products: exploring advanced techniques, carrier agents, applications, and limitations. Food and Bioprocess Technology. 18: 1181-1220. [DOI: 10.1007/s11947-024-03544-4] [DOI:10.1007/s11947-024-03544-4]
17. Xi D., Lin N., Gori A. (2023). Increasing sequential tropical cyclone hazards along the US East and Gulf coasts. Nature Climate Change. 13: 258-265. [DOI: 10.1038/s41558-023-01595-7] [DOI:10.1038/s41558-023-01595-7]
18. Yaashikaa P.R., Kamalesh R., Senthil Kumar P., Saravanan A., Vijayasri K., Rangasamy G. (2023). Recent advances in edible coatings and their application in food packaging. Food Research International. 173: 113366. [DOI: 10.1016/ j.foodres.2023.113366] [DOI:10.1016/j.foodres.2023.113366] [PMID]
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |







