Volume 12, Issue 2 (June 2025)
J. Food Qual. Hazards Control 2025, 12(2): 104-117 |
Back to browse issues page
Ethics code: Not applicable.
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Khaerani Z, Sri Palupi N, Wulandari N, Kusnandar F. Chemical Characteristics and Allergenicity Alterations Induced by Heating in Mackerel Tuna (Euthynnus affinis) Powder Production. J. Food Qual. Hazards Control 2025; 12 (2) :104-117
URL: http://jfqhc.ssu.ac.ir/article-1-1283-en.html
URL: http://jfqhc.ssu.ac.ir/article-1-1283-en.html
Department of Food Science and Technology, Faculty of Agricultural Technology, IPB University, Bogor, Indonesia, Southeast Asian Food and Agricultural Science and Technology (SEAFAST) Center, IPB University, Bogor, Indonesia, Department of Food Science and Technology, Faculty of Agricultural Engineering and Technology, IPB University, Bogor, Indonesia , hnpalupi@apps.ipb.ac.id
Full-Text [PDF 885 kb]
(373 Downloads)
| Abstract (HTML) (794 Views)
Full-Text: (168 Views)
Chemical Characteristics and Allergenicity Alterations Induced by Heating in Mackerel Tuna (Euthynnus affinis) Powder Production
Z. Khaerani 1, N. Sri Palupi 2,3,4[*]* , N. Wulandari 2,3 , F. Kusnandar 2,3
1. Food Science Study Program, Graduate School, IPB University, Bogor, Indonesia 16680
2. Department of Food Science and Technology, Faculty of Agricultural Technology, IPB University, Bogor, Indonesia
3. Southeast Asian Food and Agricultural Science and Technology (SEAFAST) Center, IPB University, Bogor, Indonesia
4. Department of Food Science and Technology, Faculty of Agricultural Engineering and Technology, IPB University, Bogor, Indonesia
HIGHLIGHTS
Table 1: Yield of mackerel tuna fish powder
Table 2: Nutritional composition of mackerel tuna during the fish powder production process
wb=Wet Base; db=Dry Base.
Different superscript letters in the same column indicate significant differences (p<0.05).

Z. Khaerani 1, N. Sri Palupi 2,3,4[*]*
1. Food Science Study Program, Graduate School, IPB University, Bogor, Indonesia 16680
2. Department of Food Science and Technology, Faculty of Agricultural Technology, IPB University, Bogor, Indonesia
3. Southeast Asian Food and Agricultural Science and Technology (SEAFAST) Center, IPB University, Bogor, Indonesia
4. Department of Food Science and Technology, Faculty of Agricultural Engineering and Technology, IPB University, Bogor, Indonesia
- Parvalbumin was the primary allergen and was present at higher levels in white meat than red meat.
- Pressure heating was more effective than steaming at diminishing both the intensity and quantity of allergenic protein bands.
- Powder production enhanced allergen reduction, achieving over 83% reduction across all meat types.
| Article type Original article |
ABSTRACT Background: Parvalbumin, the main allergen in mackerel tuna (Euthynnus affinis), is present in higher concentrations in white meat relative to red. Parvalbumin reduction can be achieved through heating methods such as pressure heating (autoclaving) and steaming. This study aimed to evaluate the impact of these methods on the nutritional value of mackerel tuna fish powder, analyze molecular weight profiles during processing, and assess allergenicity across different meat types. Methods: Fish powder production begins by separating the meat into three types: white, red, and mixed with a weight ratio of red meat to white meat of 1:3 (w/w). Fifteen samples—five per meat type, including fresh, post-heating, and post-flour processing were analyzed in January 2024, with each treatment performed in duplicate. Nutritional composition (proximate analysis), protein molecular weight (Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis), and allergenicity (Enzyme-Linked Immuno-Sorbent Assay kit) were evaluated. Statistical analysis was conducted using SPSS 25.0, with significance set at 5%. Data were analyzed using t-tests and Analysis of Variance (ANOVA). Duncan’s Multiple Range Test (DMRT) was applied to evaluate significant differences. Results: Pressure heating and steaming both significantly reduced moisture content and increased protein levels in fish powder. Pressure heating more effectively reduced allergenic protein bands, especially parvalbumin, compared to steaming. Moreover, pressure heating achieved greater allergenicity reduction in white (85.49%), red (84.24%), and mixed meats (83.01%). White meat exhibited the highest parvalbumin levels, followed by mixed and red meats. Conclusion: Both heating methods effectively reduced allergenicity. Based on allergenicity and economic factors, mixed meat types are recommended. © 2025, Shahid Sadoughi University of Medical Sciences. This is an open access article under the Creative Commons Attribution 4.0 International License. |
|
| Keywords Allergens Mackerels Perciformes Steam |
||
| Article history Received: 9 Nov 2024 Revised: 3 Feb 2025 Accepted: 21 May 2025 |
||
| Abbreviations ELISA= Enzyme-Linked Immuno-Sorbent Assay SDS-PAGE=Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis |
To cite: Khaerani Z., Sri Palupi N., Wulandari N., Kusnandar F. (2025). Chemical characteristics and allergenicity alterations induced by heating in mackerel tuna (Euthynnus affinis) powder production. Journal of Food Quality and Hazards Control. 12: 104-117.
Introduction
Introduction
Indonesia's extensive maritime territory offers significant potential for producing fishery resources. The population of Mackerel tuna (Euthynnus affinis) in Indonesian waters appears abundant, as reflected by findings from the Java sea showing wide distribution, year-round recruitment, and high catch volumes across multiple coastal areas (Mardlijah et al., 2022). The abundant supply, stable availability, and low cost of mackerel tuna make it a top-rated source of animal protein in Indonesia.
The protein content in mackerel tuna comes primarily from its meat, which constitutes nearly 50% of the fish's total mass. Mackerel tuna muscle proteins are classified into myofibrillar, stromal, and sarcoplasmic categories, with sarcoplasmic proteins accounting for 25-30% of the total muscle protein. These water-soluble proteins include myoglobin, various enzymes, and parvalbumin(Strasburg et al., 2017) .
Globally, the World Allergy Organization (WAO) reports that between 220 and 550 million people suffer from food allergies(WAO, 2013) . Fish allergy is relatively uncommon worldwide, affecting less than 1% of the population; however, its prevalence is higher in regions with high fish consumption. In areas with extensive fish-processing industries, the prevalence may increase to 3%. Among pediatric populations, fish allergy can affect up to 7% of individuals, whereas its occurrence is generally lower in adults (Mastrorilli et al., 2023). Although data on fish allergies in Indonesia is limited, a study conducted in several schools in Surabaya revealed that 0.8% of adolescents aged 13-18 and 2.66% of adults over 19 suffer from fish allergies (Soegiarto et al., 2019).
The primary cause of fish allergies, including those related to mackerel tuna, is parvalbumin, a small protein with a molecular weight of 10-12 kDa(Prester, 2016) . Allergic reactions are triggered when Immunoglobulin E (IgE) antibodies recognize peptide fragments in parvalbumin as allergenic compounds (Kamath et al., 2023). Following an allergic reaction, the body responds by releasing mediators that stimulate increased mucus production, vasodilation, and enhanced vascular permeability. These physiological changes can lead to symptoms such as swelling, abdominal discomfort, nausea, diarrhea, and skin rashes. In more severe cases, the reaction may cause bronchial constriction and anaphylaxis (Tedner et al., 2022). Both intrinsic and extrinsic factors influence the allergic response to parvalbumin. Intrinsic factors include the fish species and muscle type, each with varying levels of parvalbumin. Studies have shown dark fish meat contains 4-8 times less parvalbumin than white fish meat (Dasanayaka et al., 2020) . Extrinsic factors, such as food processing methods, also affect allergenicity. Techniques like heating, high-pressure treatment, radiation, and ultrasound can alter protein structures, affecting the allergen's conformational epitopes. Additionally, linear epitopes may degrade into smaller fragments during microbial fermentation or enzymatic hydrolysis, reducing allergenicity (Zhou et al., 2021) .
Heating processes are commonly employed to extend the shelf-life of fish products, such as in producing fish powder. Fish powder is an intermediate product easily incorporated into various food products. The fish powder production process is designed to increase protein content(Khasanah et al., 2020; Maulidah et al., 2022) . It involves several steps including washing, cooking, drying, grinding, and sieving (Maulidah et al., 2022) . Typically, cooking during fish powder production involves either boiling or steaming. However, modifying this process using pressure heating can reduce the allergenicity of the resulting fish powder. Research on white snapper, for example, has shown that autoclaving for 30 min results in lower parvalbumin levels compared to boiling for 10 min at 98 °C (Schrama et al., 2022) .
While previous studies primarily focus on allergen reduction, they often lack direct applications in hypoallergenic food development. This study goes beyond merely identifying the most effective processing method for reducing allergenicity by assessing its practical implications for fish powder formulation. By integrating allergen reduction with nutritional enhancement, this research provides valuable insights for both the food industry and consumers, bridging the gap between scientific findings and real-world applications.
Given the potential of fish powder as a hypoallergenic food ingredient, particularly for individuals with allergies, this study aimed to evaluate the effects of heating (both pressure heating and steaming) on the nutritional value during mackerel tuna fish powder production, analyze the molecular weight profile throughout the production process, and assess the level of allergenicity in mackerel tuna fish powder made from three different types of meat.
The protein content in mackerel tuna comes primarily from its meat, which constitutes nearly 50% of the fish's total mass. Mackerel tuna muscle proteins are classified into myofibrillar, stromal, and sarcoplasmic categories, with sarcoplasmic proteins accounting for 25-30% of the total muscle protein. These water-soluble proteins include myoglobin, various enzymes, and parvalbumin
Globally, the World Allergy Organization (WAO) reports that between 220 and 550 million people suffer from food allergies
The primary cause of fish allergies, including those related to mackerel tuna, is parvalbumin, a small protein with a molecular weight of 10-12 kDa
Heating processes are commonly employed to extend the shelf-life of fish products, such as in producing fish powder. Fish powder is an intermediate product easily incorporated into various food products. The fish powder production process is designed to increase protein content
While previous studies primarily focus on allergen reduction, they often lack direct applications in hypoallergenic food development. This study goes beyond merely identifying the most effective processing method for reducing allergenicity by assessing its practical implications for fish powder formulation. By integrating allergen reduction with nutritional enhancement, this research provides valuable insights for both the food industry and consumers, bridging the gap between scientific findings and real-world applications.
Given the potential of fish powder as a hypoallergenic food ingredient, particularly for individuals with allergies, this study aimed to evaluate the effects of heating (both pressure heating and steaming) on the nutritional value during mackerel tuna fish powder production, analyze the molecular weight profile throughout the production process, and assess the level of allergenicity in mackerel tuna fish powder made from three different types of meat.
Materials and methods
Materials
The main ingredient in this study was mackerel tuna meat obtained from Muara Angke, North Jakarta in 2024. The materials used for analysis included Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE) solution: ammonium persulfate (APS; (CAS 7727-54-0, Sigma-Aldrich, Germany), tris-base (77-86-1, Sigma-Aldrich, Germany), glacial acetic acid (CAS 64-19-7, Merck, Germany), tween-20 (CAS No.9005-64-5, Sigma, USA), Tetramethylethylenediamine (TEMED; CAS 110-18-9, Merck, Germany), sodium dodecyl sulphate (SDS; MSDS–817034, Merck, Germany), bis-acrylamide solution (30%, 29:1) (1610156, BIORAD, UK), pectra multicolor broad range protein ladder (26634, Thermo Scientific, USA), and fish protein Enzyme-Linked Immuno-Sorbent Assay Kit (ELISA; E96FSH, 3MTM, USA). The equipments used to make fish powder consist of autoclave (Korimat, Europe), steamer (Armfield, UK), drum dryer (Tummers Simon Dryers Technology, UK), grinder (GEA Getra, Indonesia), and an 80 mesh sieve. Furthermore, centrifuges (Hermle, Germany), SDS-PAGE units (BIORAD, UK), and microplate reader (BIORAD, UK) were utilized to support research and analysis.
Mackerel tuna powder production
Fish powder production begins by separating the meat into three types: white, red, and mixed with a weight ratio of red meat to white meat of 1:3 (w/w). Furthermore, a heating process is carried out consisting of two steps: (1) pressure heating at 121 °C for 30 min(Maulidah et al., 2022) and (2) steaming at 100 °C for 30 min (Khasanah et al., 2020) . The fish meat is then dried using a drum dryer (Pramestia et al., 2015) at 148 °C, p=5 bar for six s/cycle. The samples are then ground using a grinder and sieved with an 80 mesh sieve. The powdered samples are then separated. Thus, each type of meat has five samples; one without process, two samples after heating, and two samples after powder production. Therefore, there are a total of 15 samples, which are then extracted and analyzed.
Sample extraction
The fat removal was performed on the fish powder samples before extraction(Wulan et al., 2024) . The fish powder samples were mixed with hexane solvent at a ratio of 1:3 (w/v) for one h at 25 °C in a water bath shaker. The process was repeated twice with the same steps. Subsequently, the solid was dried in an oven at 50 °C for one h. Protein extraction was carried out using Tris-Buffered Saline (TBS; 20 mm Tris-HCl, 150 mm NaCl, pH 7.4) solvent overnight at four °C at a ratio of 1:5 (w/v). The mixture was then centrifuged at 10,000 × g for 30 min. The supernatant was filtered through filter paper and stored at −20 °C for analysis (Nugraha et al., 2021; Palupi et al., 2021) .
Yield
The yield of mackerel tuna fish powder is calculated by comparing the final weight (dry fish powder) with the initial weight of fresh meat mackerel tuna fish. These measurements are used to determine the efficiency of the mackerel tuna fish powder manufacturing process.
Nutritional composition analysis
The nutritional composition of the samples was carried out using a proximate test in accordance with AOAC International (1995). This analysis encompassed water content (oven method), ash content (gravimetric method), fat content (Soxhlet method), and protein content (Kjedhal method). Carbohydrate content was calculated by difference..
Protein molecular weight by SDS-PAGE
Protein molecular weight analysis with SDS-PAGE was carried out using gel with concentration stacking gel 5% and separating gel 12%. A 12 μl sample was injected into the well, streaming the current with a voltage of 100 volts for 90 min. The gel was colored with solution staining for 30 min and washed using solution destaining (Laemmli, 1970). The molecular weight and intensity of the protein bands were determined using Gel Analyzer 23.1 software.
Allergenicity analysis with ELISA kit
The test was conducted using the fish protein ELISA Kit (3MTM, USA) based on the principle of sandwich ELISA. The sample, extracted with the extraction buffer, was injected into the ELISA well containing the primary antibody. The sample was then analyzed using methods and reagents included in the kit. Absorbance was measured at a length of 450 nm wave using a microplate reader (BIORAD, UK)(Abbott et al., 2010) .
Statistical analysis
Data were analyzed using t-tests and ANOVA for white, red, and mixed meat under a Completely Randomized Design. Each treatment performed in duplicate. Statistical analysis was conducted using MS Excel 365 and SPSS 25.0, with significance set at 5%. Duncan’s Multiple Range Test (DMRT) was applied to identify significant differences.
Results and Discussion
Yield
The yield was utilized to assess the efficiency of mackerel tuna meat utilization in producing fish powder. The heating and drying process (Table 1) produced a yield: 18% for white meat, 16-17% for red meat, and 20% for mixed meat. The yield after drying showed relatively similar values between pressure heating and steaming across all three types of meat (white, red, and mixed). Statistically, the p-value indicated no significant difference between the two heating methods (p>0.05). These values represent a reduction to approximately 80% of the yield after heating. This decline can be attributed to water loss during heating and milling. A reduction in the yield of the dried product was noted as the drying temperature increased. This may be attributed to a greater loss of moisture during the drying process, resulting in a lower final mass of the dried material (Tontul and Topuz, 2017).
The fish powder yield from mixed meat is more economically advantageous than that from white meat because mixed meat includes red meat, which is often underutilized and considered waste in processed products. Incorporating red meat increases the product’s economic value(Li et al., 2017) . Optimizing these processing by-products can boost total production by up to 61% (Stevens et al., 2018) .
Materials
The main ingredient in this study was mackerel tuna meat obtained from Muara Angke, North Jakarta in 2024. The materials used for analysis included Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE) solution: ammonium persulfate (APS; (CAS 7727-54-0, Sigma-Aldrich, Germany), tris-base (77-86-1, Sigma-Aldrich, Germany), glacial acetic acid (CAS 64-19-7, Merck, Germany), tween-20 (CAS No.9005-64-5, Sigma, USA), Tetramethylethylenediamine (TEMED; CAS 110-18-9, Merck, Germany), sodium dodecyl sulphate (SDS; MSDS–817034, Merck, Germany), bis-acrylamide solution (30%, 29:1) (1610156, BIORAD, UK), pectra multicolor broad range protein ladder (26634, Thermo Scientific, USA), and fish protein Enzyme-Linked Immuno-Sorbent Assay Kit (ELISA; E96FSH, 3MTM, USA). The equipments used to make fish powder consist of autoclave (Korimat, Europe), steamer (Armfield, UK), drum dryer (Tummers Simon Dryers Technology, UK), grinder (GEA Getra, Indonesia), and an 80 mesh sieve. Furthermore, centrifuges (Hermle, Germany), SDS-PAGE units (BIORAD, UK), and microplate reader (BIORAD, UK) were utilized to support research and analysis.
Mackerel tuna powder production
Fish powder production begins by separating the meat into three types: white, red, and mixed with a weight ratio of red meat to white meat of 1:3 (w/w). Furthermore, a heating process is carried out consisting of two steps: (1) pressure heating at 121 °C for 30 min
Sample extraction
The fat removal was performed on the fish powder samples before extraction
Yield
The yield of mackerel tuna fish powder is calculated by comparing the final weight (dry fish powder) with the initial weight of fresh meat mackerel tuna fish. These measurements are used to determine the efficiency of the mackerel tuna fish powder manufacturing process.
Nutritional composition analysis
The nutritional composition of the samples was carried out using a proximate test in accordance with AOAC International (1995). This analysis encompassed water content (oven method), ash content (gravimetric method), fat content (Soxhlet method), and protein content (Kjedhal method). Carbohydrate content was calculated by difference..
Protein molecular weight by SDS-PAGE
Protein molecular weight analysis with SDS-PAGE was carried out using gel with concentration stacking gel 5% and separating gel 12%. A 12 μl sample was injected into the well, streaming the current with a voltage of 100 volts for 90 min. The gel was colored with solution staining for 30 min and washed using solution destaining (Laemmli, 1970). The molecular weight and intensity of the protein bands were determined using Gel Analyzer 23.1 software.
Allergenicity analysis with ELISA kit
The test was conducted using the fish protein ELISA Kit (3MTM, USA) based on the principle of sandwich ELISA. The sample, extracted with the extraction buffer, was injected into the ELISA well containing the primary antibody. The sample was then analyzed using methods and reagents included in the kit. Absorbance was measured at a length of 450 nm wave using a microplate reader (BIORAD, UK)
Statistical analysis
Data were analyzed using t-tests and ANOVA for white, red, and mixed meat under a Completely Randomized Design. Each treatment performed in duplicate. Statistical analysis was conducted using MS Excel 365 and SPSS 25.0, with significance set at 5%. Duncan’s Multiple Range Test (DMRT) was applied to identify significant differences.
Results and Discussion
Yield
The yield was utilized to assess the efficiency of mackerel tuna meat utilization in producing fish powder. The heating and drying process (Table 1) produced a yield: 18% for white meat, 16-17% for red meat, and 20% for mixed meat. The yield after drying showed relatively similar values between pressure heating and steaming across all three types of meat (white, red, and mixed). Statistically, the p-value indicated no significant difference between the two heating methods (p>0.05). These values represent a reduction to approximately 80% of the yield after heating. This decline can be attributed to water loss during heating and milling. A reduction in the yield of the dried product was noted as the drying temperature increased. This may be attributed to a greater loss of moisture during the drying process, resulting in a lower final mass of the dried material (Tontul and Topuz, 2017).
The fish powder yield from mixed meat is more economically advantageous than that from white meat because mixed meat includes red meat, which is often underutilized and considered waste in processed products. Incorporating red meat increases the product’s economic value
Table 1: Yield of mackerel tuna fish powder
| Treatment | Fresh meat weight (g) | Yield (%) | |||
| post heating | p-value | post drying | p-value | ||
| White meat | |||||
| Pressure heating | 562.50 | 62.05±1.02 | 0.081 | 18.25±1.36 | 0.858 |
| Steaming | 539.55 | 67.36±2.04 | 18.32±1.47 | ||
| Red meat | |||||
| Pressure heating | 182.40 | 60.70±3.06 | 0.155 | 16.10±4.58 | 0.563 |
| Steaming | 191.15 | 69.63±4.77 | 17.64±3.37 | ||
| Mixed meat | |||||
| Pressure heating | 742.75 | 63.50±1.12 | 0.055 | 20.66±0.22 | 0.551 |
| Steaming | 723.65 | 67.22±0.64 | 20.76±0.49 | ||
Nutritional composition
The processing steps, including pressure heating, steaming, and powder production, significantly impacted the nutritional profile of the final product, namely fish powder. The nutritional values (Table 2) showed a substantial reduction in moisture content and increased protein levels. These changes indicate a concentration of nutrients, which can be beneficial for certain applications. However, only minor changes were observed in ash, fat, and carbohydrate contents, which may not significantly affect the nutritional quality of the fish powder.
Moisture content decreased following the heating process (pressure heating and steaming) and powder production (drying and milling). The initial water content of 71.98±1.16-73.29±1.35% was reduced to 61.13±0.31-62.18±0.55% after pressure heating and to 63.48±0.20-66.65±0.81% after steaming. Cooking the fish using these methods causes the release of free water from the food, lowering its water content(Adawyah et al., 2020) . The drying process similarly affected the removal of free water from the food. A decrease in moisture content of 3.34±0.11-6.69±0.27% was observed in the fish powder, caused by direct contact between the fish and the hot surface of the drum dryer, which accelerated water evaporation. The longer the material remained in contact with the drum dryer (due to the lower drum rotation speed), the more significant the reduction in moisture content (Rittisak et al., 2022) . In terms of meat type (fresh or processed), white meat exhibited a higher moisture content compared to red and mixed meat, which is consistent with previous research (Kannaiyan et al., 2019) , showing that white mackerel tuna meat had a higher moisture content (75.52%) than red meat (74.85%).
Ash content in food reflects the presence of minerals and inorganic compounds. Pressure heating and steaming, can reduce ash content by affecting the mineral composition of the material. Ash content ranged from 4.62±0.49-5.71±0.61% in fresh products, 2.66±0.19-3.93±0.56% after pressure heating, 3.99±0.09-4.41±0.75% after steaming, and 3.36±0.18-4.70±0.02% after powder production. The reduction in ash content following heating may attribute to the loss of dissolved minerals through evaporation. Meanwhile, a study by Kong et al. (2024) indicates that ash content decreases under pressure cooking treatments and increases under steaming treatments. In Table 2, the ash content decreases after heating but increases again following the powder production process (except in red meat). This increase in ash content after powder production demonstrates a significant effect influenced by meat type and heating method. The average ash content in flour processed by pressure heating and steaming is 4.02±0.58 and 3.86±0.33%, respectively. This increase is attributed to the reduction in moisture content, leading to a relative rise in ash concentration compared to the total mass of the material (Ariva et al., 2020). The ash content in fish powder can increase up to fourfold compared to its fresh form(Tiwari et al., 2021) . White meat generally had lower ash content than red and mixed meat (except in the steaming process). The higher ash content in red meat is likely due to its higher concentrations of minerals such as iron (Fe), cupper (Cu), potassium (K), magnesium (Mg), and calcium (Ca) (Albrecht-Ruiz and Salas-Maldonado, 2015) .
Fat content changed significantly after pressure heating but did not show notable differences after steaming and powder production. The fat content of fresh fish, which ranged from 1.37±0.46-2.91±1.82%, increased to 7.40±1.41-8.97±0.30% after pressure heating. This increase may be attributed to changes in the composition of saturated and unsaturated fatty acids due to the effects of heat and pressure. These processes can trigger triglyceride hydrolysis, leading to higher levels of free fatty acids(Lakmini et al., 2022) . The polyunsaturated fatty acid content in mackerel tuna, particularly omega-3 and omega-6, also demonstrated more excellent stability after pressure heating than steaming and grilling (Kilic, 2022) . Similar findings were observed in a study of swamp gourami, where pressure-heated fish exhibited higher fat content than steamed fish (Adawyah et al., 2020) . Regarding meat types, red meat (fresh, pressure-heated, and pressure-heated-powdered) contained higher fat levels than white and mixed meats, which aligns with research showing that red meat had triple the total fat content of white meat and a higher proportion of polyunsaturated fatty acids (Kannaiyan et al., 2019) . However, white meat showed an increase in fat content after steaming and steaming-powder production, potentially due to the instability of free fatty acids in red meat following steaming, which did not significantly affect other meat types.
The processing steps, including pressure heating, steaming, and powder production, significantly impacted the nutritional profile of the final product, namely fish powder. The nutritional values (Table 2) showed a substantial reduction in moisture content and increased protein levels. These changes indicate a concentration of nutrients, which can be beneficial for certain applications. However, only minor changes were observed in ash, fat, and carbohydrate contents, which may not significantly affect the nutritional quality of the fish powder.
Moisture content decreased following the heating process (pressure heating and steaming) and powder production (drying and milling). The initial water content of 71.98±1.16-73.29±1.35% was reduced to 61.13±0.31-62.18±0.55% after pressure heating and to 63.48±0.20-66.65±0.81% after steaming. Cooking the fish using these methods causes the release of free water from the food, lowering its water content
Ash content in food reflects the presence of minerals and inorganic compounds. Pressure heating and steaming, can reduce ash content by affecting the mineral composition of the material. Ash content ranged from 4.62±0.49-5.71±0.61% in fresh products, 2.66±0.19-3.93±0.56% after pressure heating, 3.99±0.09-4.41±0.75% after steaming, and 3.36±0.18-4.70±0.02% after powder production. The reduction in ash content following heating may attribute to the loss of dissolved minerals through evaporation. Meanwhile, a study by Kong et al. (2024) indicates that ash content decreases under pressure cooking treatments and increases under steaming treatments. In Table 2, the ash content decreases after heating but increases again following the powder production process (except in red meat). This increase in ash content after powder production demonstrates a significant effect influenced by meat type and heating method. The average ash content in flour processed by pressure heating and steaming is 4.02±0.58 and 3.86±0.33%, respectively. This increase is attributed to the reduction in moisture content, leading to a relative rise in ash concentration compared to the total mass of the material (Ariva et al., 2020). The ash content in fish powder can increase up to fourfold compared to its fresh form
Fat content changed significantly after pressure heating but did not show notable differences after steaming and powder production. The fat content of fresh fish, which ranged from 1.37±0.46-2.91±1.82%, increased to 7.40±1.41-8.97±0.30% after pressure heating. This increase may be attributed to changes in the composition of saturated and unsaturated fatty acids due to the effects of heat and pressure. These processes can trigger triglyceride hydrolysis, leading to higher levels of free fatty acids
Table 2: Nutritional composition of mackerel tuna during the fish powder production process
| Treatment/ Sample |
Water Content (wb%) | Ash Content (db%) | Fat Content (db%) | Protein Content (db%) | Carbohydrate Content (db%) |
| White meat | |||||
| Fresh white meat | 73.26±1.73 a | 4.62±0.49 a | 1.93±0.52 c | 86.07±1.28 a | 7.39±5.29 a |
| Autoclaved white meat | 61.50±1.01 c | 2.66±0.19 c | 7.40±1.41 a | 89.15±0.84 b | 1.15±1.26 b |
| Steamed white meat | 65.38±0.40 b | 4.41±0.75 a | 4.76±1.48 b | 89.11±2.79 b | 2.23±1.32 ab |
| Autoclaved white meat powder | 5.09±0.46 d | 3.36±0.18 b | 2.59±0.17 c | 92.37±1.11 c | 5.96±5.64 ab |
| Steamed white meat powder | 6.41±0.22 d | 3.49±0.09 b | 3.14±0.56 c | 92.48±0.42 c | 3.14±2.46 ab |
| Red meat | |||||
| Fresh red meat | 71.98±1.16 a | 5.15±0.18 a | 2.91±1.82 b | 76.23±1.45 a | 16.48±1.62 a |
| Autoclaved red meat | 61.13±0.31 c | 3.72±0.41 d | 8.97±0.30 a | 86.68±0.46 b | 0.92±1.09 b |
| Steamed red meat | 63.48±0.20 b | 3.99±0.09 cd | 1.79±0.29 b | 93.30±1.06 d | 1.27±1.03 b |
| Autoclaved red meat powder | 3.73±0.18 d | 4.70±0.02 c | 2.81±0.19 b | 89.82±2.94 c | 9.60±10.77 a |
| Steamed red meat powder | 3.34±0.11 d | 4.24±0.08 b | 2.23±0.21 b | 91.00±0.65 cd | 9.15±2.50 a |
| Mixed meat | |||||
| Fresh mixed meat | 73.29±1.35 a | 5.71±0.61 a | 1.37±0.46 a | 78.38±1.19 a | 14.53±5.70 a |
| Autoclaved mixed meat | 62.18±0.55 c | 3.93±0.56 b | 7.80±0.78 d | 86.47±3.14 b | 2.53±2.81 bc |
| Steamed mixed meat | 66.65±0.81 b | 4.37±0.18 b | 1.96±0.65 ab | 89.04±2.42 b | 5.79±1.61 b |
| Autoclaved mixed meat powder | 5.67±0.09 d | 3.99±0.10 b | 2.66±0.44 bc | 93.24±0.27 c | 0.40±0.48 c |
| Steamed mixed meat powder | 6.69±0.27 d | 3.87±0.33 b | 3.02±0.52 c | 92.44±0.64 c | 2.35±1.10 bc |
Different superscript letters in the same column indicate significant differences (p<0.05).
The method of processing fresh mackerel tuna into fish powder significantly impacted protein content. The protein content of fresh mackerel tuna ranged from 76.23±1.45 to 86.07±1.28%, which increased to 86.47±3.14 to 89.15±0.84% after pressure heating, 89.04±2.42 to 93.30±1.06% after steaming, and 89.82±2.94 to 93.24±0.27% after the powder production process. These changes in protein content have significant implications for the nutritional value and quality of processed mackerel tuna. Previous research on salmon and mackerel demonstrated a significant increase in protein content following steaming and canning using an autoclave (Bastías et al., 2017) . This rise in protein content is attributed to reduced water content during heat treatment, leading to protein solids concentration, including collagen, muscle tissue fragments, sarcoplasmic proteins, and fat (Bastías et al., 2017) .
The increase in protein content observed after drying is not solely due to moisture reduction but also to the retention of nitrogen compounds including protein and Non-Protein Nitrogen (NPN). As water content decreases in food, the relative concentration of nitrogen compounds, such as proteins and their degradation products, increases. The drying process may also initiate the breakdown of proteins into peptides and free amino acids, thereby increasing NPN content, such as ammonia, trimethylamine (TMA), and dimethylamine (DMA)(Nguyen et al., 2022) . Other studies have also reported a gradual increase in protein content during drying, attributed to the dehydration of water molecules between proteins, which promotes protein aggregation (Akhtara and Borah, 2022) . Regarding meat types, white mackerel tuna meat generally has a higher protein content than red or mixed meat. However, other studies indicate that the protein content between red and white meat is not significantly different, with both types containing nearly equivalent protein levels at approximately 23.12±0.13% for white meat and 23.15±0.02% for red meat (Kannaiyan et al., 2019) .
Protein molecular weight profile by SDS-PAGE
The molecular weight profile of fresh and processed mackerel tuna proteins was examined using SDS-PAGE. Based on their molecular weight, the separation of protein bands on a polyacrylamide gel revealed a protein profile, as shown in Figure 1. According to previous studies(Ruethers et al., 2018) , several protein bands have been identified as allergenic in fish including parvalbumin (10-13 kDa), tropomyosin (33-39 kDa), aldolase A (40 kDa), β-enolase (50 kDa), and collagen (>100 kDa). In addition to allergenic proteins, other proteins commonly found in fish include Myosin Heavy Chain (MHC) (221.6 kDa), Myosin Light Chain (MLC) (16.8 kDa), actin (42 kDa), troponin subunit T (33.6 kDa), troponin subunit I (19.7 kDa), troponin subunit C (18.2 kDa), paramyosin (107.5 kDa), α-actinin (103.9 kDa), and myoglobin (15.6 kDa) (Ochiai and Ozawa, 2020) .
Figure 1 displays differences in the number and intensity of protein bands detected in fresh mackerel tuna including white, red, and mixed meat. Specifically, 14 protein bands were identified in white meat, 17 in red meat, and 13 in mixed meat. Darker shades marked protein bands with higher intensities. Non-allergenic protein bands detected in fresh mackerel tuna included those at 16-17 kDa (MLC) in white and mixed meat, 33 kDa (troponin subunit T) in all meat types, 15-16 kDa (myoglobin) in red and mixed meat, and 19 kDa (troponin subunit I) in red meat. Conversely, protein bands at 10-13 kDa (parvalbumin) and 33-39 kDa (tropomyosin) were suspected to be allergenic.
Following heat treatments, including pressure heating, steaming, and powder production, changes in the number and intensity of allergenic protein bands, specifically parvalbumin and tropomyosin, were observed. These changes are shown in Figure 2. In white meat, the number of protein bands decreased after heat treatment. The initial 14 protein bands in fresh meat were reduced to nine after pressure heating and to 10 after steaming, but increased again to 13 after the powder production process. In addition to band number changes, intensity variations were noted. Pressure heating led to a 100% reduction in parvalbumin intensity and a 75% reduction in tropomyosin intensity. In comparison, steaming resulted in a more than 100% increase in parvalbumin intensity and a 62% decrease in tropomyosin intensity.
Fresh red meat had 17 protein bands, but its lower stability resulted in a loss of many bands after processing. Following treatment, the bands detected were seven, four, three, and seven for pressure heating, steaming, powder-pressure heating, and powder-steaming, respectively. This band reduction was accompanied by decreased intensity, particularly in parvalbumin, which increased the 12 kDa band after steaming.
In mixed meat, the number of protein bands initially stood at 13. After undergoing pressure heating, steaming, powder-pressure heating, and powder-steaming, the bands changed to four, six, nine, and 12, respectively. However, the large number of protein bands after the combined steaming and powder production was directly proportional to the increased intensity of parvalbumin.
Pressure heating effectively reduced the number and intensity of protein bands, particularly parvalbumin and tropomyosin. In contrast, steaming tended to increase the number and intensity of bands. The increase in band intensity is presumably a result of heating, which leads to the unfolding of peptide chains, thereby gradually exposing more hidden epitopes (Chen and Hsieh, 2021). After powder production, the number and intensity of protein bands rose again. The reduction in protein bands during pressure heating was attributed to the combined effects of heat and pressure, which caused the secondary structure of parvalbumin, such as α-helix and β-strands, to decrease by up to two-fold, resulting in more irregular coils(De Jongh et al., 2015) . Heating at 60-100 ˚C eliminated certain protein bands, such as those at 37 and 50 kDa, while boiling at 120-140 ˚C effectively reduced bands in the 10-100 kDa range in salmon (Kubota et al., 2016) . Heating at 100 ˚C for 30 min also reduced the intensity of bands at 10 and 35 kDa compared to unheated samples (Dasanayaka et al., 2022) . Similarly, studies on tilapia (Oreochromis niloticus) and mud grouper (Epinephelus coioides) found that parvalbumin (10 kDa) and tropomyosin (35 kDa) bands remained detectable after heat treatments involving steaming (10 min) and autoclaving (20 min) (Tsai et al., 2023) .
In general, red meat exhibits more protein bands when fresh compared to other types of meat. However, after heating, the number of protein bands in red meat decreases significantly due to its lower stability during processing and storage(Li et al., 2017) . This phenomenon is also observed in the sarcoplasmic proteins of carp, where red meat shows a more significant number of protein bands, although with lower intensity than white meat (Li et al., 2016) . SDS-PAGE analysis of sarcoplasmic proteins in carp revealed that white meat contained a total of 22 protein bands with a range of molecular weights, including 11 major protein bands at 12 kDa, three bands in the 22-35 kDa range, two bands between 35-40 kDa, and individual bands at 50, 60, 100, and 130-180 kDa. In contrast, red meat had only six detectable protein bands, consisting of three bands within the 12-15 kDa range, one between 35-40 kDa, one between 40-55 kDa, and one at 180 kDa (Li et al., 2017) .
The increase in protein content observed after drying is not solely due to moisture reduction but also to the retention of nitrogen compounds including protein and Non-Protein Nitrogen (NPN). As water content decreases in food, the relative concentration of nitrogen compounds, such as proteins and their degradation products, increases. The drying process may also initiate the breakdown of proteins into peptides and free amino acids, thereby increasing NPN content, such as ammonia, trimethylamine (TMA), and dimethylamine (DMA)
Protein molecular weight profile by SDS-PAGE
The molecular weight profile of fresh and processed mackerel tuna proteins was examined using SDS-PAGE. Based on their molecular weight, the separation of protein bands on a polyacrylamide gel revealed a protein profile, as shown in Figure 1. According to previous studies
Figure 1 displays differences in the number and intensity of protein bands detected in fresh mackerel tuna including white, red, and mixed meat. Specifically, 14 protein bands were identified in white meat, 17 in red meat, and 13 in mixed meat. Darker shades marked protein bands with higher intensities. Non-allergenic protein bands detected in fresh mackerel tuna included those at 16-17 kDa (MLC) in white and mixed meat, 33 kDa (troponin subunit T) in all meat types, 15-16 kDa (myoglobin) in red and mixed meat, and 19 kDa (troponin subunit I) in red meat. Conversely, protein bands at 10-13 kDa (parvalbumin) and 33-39 kDa (tropomyosin) were suspected to be allergenic.
Following heat treatments, including pressure heating, steaming, and powder production, changes in the number and intensity of allergenic protein bands, specifically parvalbumin and tropomyosin, were observed. These changes are shown in Figure 2. In white meat, the number of protein bands decreased after heat treatment. The initial 14 protein bands in fresh meat were reduced to nine after pressure heating and to 10 after steaming, but increased again to 13 after the powder production process. In addition to band number changes, intensity variations were noted. Pressure heating led to a 100% reduction in parvalbumin intensity and a 75% reduction in tropomyosin intensity. In comparison, steaming resulted in a more than 100% increase in parvalbumin intensity and a 62% decrease in tropomyosin intensity.
Fresh red meat had 17 protein bands, but its lower stability resulted in a loss of many bands after processing. Following treatment, the bands detected were seven, four, three, and seven for pressure heating, steaming, powder-pressure heating, and powder-steaming, respectively. This band reduction was accompanied by decreased intensity, particularly in parvalbumin, which increased the 12 kDa band after steaming.
In mixed meat, the number of protein bands initially stood at 13. After undergoing pressure heating, steaming, powder-pressure heating, and powder-steaming, the bands changed to four, six, nine, and 12, respectively. However, the large number of protein bands after the combined steaming and powder production was directly proportional to the increased intensity of parvalbumin.
Pressure heating effectively reduced the number and intensity of protein bands, particularly parvalbumin and tropomyosin. In contrast, steaming tended to increase the number and intensity of bands. The increase in band intensity is presumably a result of heating, which leads to the unfolding of peptide chains, thereby gradually exposing more hidden epitopes (Chen and Hsieh, 2021). After powder production, the number and intensity of protein bands rose again. The reduction in protein bands during pressure heating was attributed to the combined effects of heat and pressure, which caused the secondary structure of parvalbumin, such as α-helix and β-strands, to decrease by up to two-fold, resulting in more irregular coils
In general, red meat exhibits more protein bands when fresh compared to other types of meat. However, after heating, the number of protein bands in red meat decreases significantly due to its lower stability during processing and storage
(a)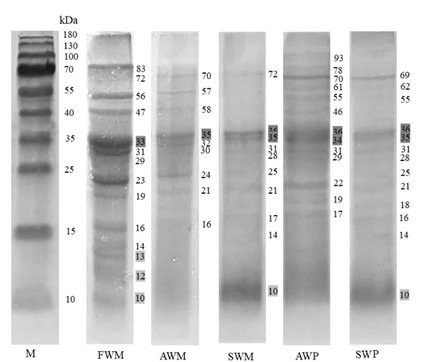 (b) .PNG) .PNG) (C) .PNG) |
.PNG) .PNG) |
Figure 1: Molecular weight profile and bands intensity of mackerel tuna during the fish powder production process in (a): white meat; (b): red meat; and (c): mixed meat
M=Marker; FWM=Fresh White Meat; AWM=Autoclaved White Meat; SWM=Steamed White Meat; AWP=Autoclaved White Meat Powder; SWP=Steamed White Meat Powder; FRM=Fresh Red Meat; ARM=Autoclaved Red Meat; SRM=Steamed Red Meat; ARP=Autoclaved Red Meat Powder; SRP=Steamed Red Meat Powder; FMM=Fresh Mixed Meat; AMM=Autoclaved Mixed Meat; SMM=Steamed Mixed Meat; AMP=Autoclaved Mixed Meat Powder; SMP=Steamed Mixed Meat Powder
M=Marker; FWM=Fresh White Meat; AWM=Autoclaved White Meat; SWM=Steamed White Meat; AWP=Autoclaved White Meat Powder; SWP=Steamed White Meat Powder; FRM=Fresh Red Meat; ARM=Autoclaved Red Meat; SRM=Steamed Red Meat; ARP=Autoclaved Red Meat Powder; SRP=Steamed Red Meat Powder; FMM=Fresh Mixed Meat; AMM=Autoclaved Mixed Meat; SMM=Steamed Mixed Meat; AMP=Autoclaved Mixed Meat Powder; SMP=Steamed Mixed Meat Powder

Figure 2: Changes in allergen band intensity of mackerel tuna allergen during the fish powder production process
Allergenicity analysis with ELISA kit
The processing of fresh mackerel tuna into powder through heating and milling stages substantially affects allergen level reduction. The changes in allergenicity were evaluated using the fish ELISA kit with the sandwich ELISA method, as shown in Figure 3. Heating, either by pressure heating or steaming for 30 min, reduced allergen levels by 68.00-74.30 and 58.91–67.32%, respectively. This reduction became more pronounced following the powder production process, which involved drying at 80 ˚C and grinding, with allergen levels decreasing by 83.01-85.49% after pressure heating and 67.45-74.55% after steaming. Overall, pressure heating proved to be more effective at lowering allergen levels than steaming, and its effectiveness was enhanced when combined with powder production. Similar findings were reported in cod, where parvalbumin levels dropped significantly after heating, from 388.2 to 29.0 µg/ml (heating at 100 ˚C) and 0.5 µg/ml (heating at 120 ˚C)(De Jongh et al., 2015) . This decrease suggests that heat can disrupt epitopes, impairing antibody binding. Epitopes in heat-resistant fractions are typically linear, while conformational epitopes from folded structures are not recognized by PoAb (Dasanayaka et al., 2022) . Additionally, heating may generate new epitopes (no allergens) or degrade existing ones, ultimately altering protein allergenicity (Laly et al., 2022) .
In addition to heating, the drying process plays a crucial role in reducing allergen levels in food products. Drying using a drum dryer has been proven effective in lowering allergenicity. A study on shrimp demonstrated that fluidized-bed drying at 45°C for 4-5 h could reduce allergen levels by up to 92% (Wulan et al., 2024). This significant reduction is believed to be associated with the prolonged drying duration, which affects the stability of allergenic protein structures. According to Al Sailawi et al. (2020), the most effective drying method for reducing allergenicity in crabs is microwave drying, followed by oven drying, sun drying, salted fish drying, and freeze drying.
Grinding also contributes to allergen reduction through significant mechanical effects. According to Vanga et al. (2017), the grinding process generates high shear stress and velocity gradients, which can break protein bonds and cause molecular structure fragmentation. This effect reduces the protein’s ability to interact with antibodies, thereby lowering its allergenicity potential.
In terms of meat type, white meat exhibited the highest parvalbumin levels compared to mixed and red meat, with concentrations of 6.61, 3.26, and 1.67 mg/g protein, respectively, in fresh samples. These results align with studies on Pacific mackerel, where parvalbumin levels were 1.86 mg/g in white meat and 0.74 mg/g in red meat(Kobayashi et al., 2016) . The elevated parvalbumin content in white meat is attributed to the greater demand for muscle relaxation during movement, a process in which parvalbumin plays a crucial role, compared to red meat (Kuniyoshi et al., 2019) . Furthermore, lower allergen levels influence the mechanism by which IgE recognizes protein epitopes in terms of linear amino acid sequences and specific three-dimensional conformational structures. These structural changes ultimately affect the immunogenicity or the ability of the proteins to induce allergic reactions (Dasanayaka et al., 2022) .
The processing of fresh mackerel tuna into powder through heating and milling stages substantially affects allergen level reduction. The changes in allergenicity were evaluated using the fish ELISA kit with the sandwich ELISA method, as shown in Figure 3. Heating, either by pressure heating or steaming for 30 min, reduced allergen levels by 68.00-74.30 and 58.91–67.32%, respectively. This reduction became more pronounced following the powder production process, which involved drying at 80 ˚C and grinding, with allergen levels decreasing by 83.01-85.49% after pressure heating and 67.45-74.55% after steaming. Overall, pressure heating proved to be more effective at lowering allergen levels than steaming, and its effectiveness was enhanced when combined with powder production. Similar findings were reported in cod, where parvalbumin levels dropped significantly after heating, from 388.2 to 29.0 µg/ml (heating at 100 ˚C) and 0.5 µg/ml (heating at 120 ˚C)
In addition to heating, the drying process plays a crucial role in reducing allergen levels in food products. Drying using a drum dryer has been proven effective in lowering allergenicity. A study on shrimp demonstrated that fluidized-bed drying at 45°C for 4-5 h could reduce allergen levels by up to 92% (Wulan et al., 2024). This significant reduction is believed to be associated with the prolonged drying duration, which affects the stability of allergenic protein structures. According to Al Sailawi et al. (2020), the most effective drying method for reducing allergenicity in crabs is microwave drying, followed by oven drying, sun drying, salted fish drying, and freeze drying.
Grinding also contributes to allergen reduction through significant mechanical effects. According to Vanga et al. (2017), the grinding process generates high shear stress and velocity gradients, which can break protein bonds and cause molecular structure fragmentation. This effect reduces the protein’s ability to interact with antibodies, thereby lowering its allergenicity potential.
In terms of meat type, white meat exhibited the highest parvalbumin levels compared to mixed and red meat, with concentrations of 6.61, 3.26, and 1.67 mg/g protein, respectively, in fresh samples. These results align with studies on Pacific mackerel, where parvalbumin levels were 1.86 mg/g in white meat and 0.74 mg/g in red meat
.PNG) |
.PNG) |
.PNG) |
Figure 3: Allergen content and percentage reduction of mackerel tuna allergen during the fish powder production process in (a): white meat; (b): red meat; and (c): mixed meat an
Different superscript letters indicate significant differences (p<0.05).
FWM=Fresh White Meat; AWM=Autoclaved White Meat; SWM=Steamed White Meat; AWP=Autoclaved White Meat Powder; SWP=Steamed White Meat Powder; FRM=Fresh Red Meat; ARM=Autoclaved Red Meat; SRM=Steamed Red Meat; ARP=Autoclaved Red Meat Powder; SRP=Steamed Red Meat Powder; FMM=Fresh Mixed Meat; AMM=Autoclaved Mixed Meat; SMM=Steamed Mixed Meat; AMP=Autoclaved Mixed Meat Powder; SMP=Steamed Mixed Meat Powder.
Different superscript letters indicate significant differences (p<0.05).
FWM=Fresh White Meat; AWM=Autoclaved White Meat; SWM=Steamed White Meat; AWP=Autoclaved White Meat Powder; SWP=Steamed White Meat Powder; FRM=Fresh Red Meat; ARM=Autoclaved Red Meat; SRM=Steamed Red Meat; ARP=Autoclaved Red Meat Powder; SRP=Steamed Red Meat Powder; FMM=Fresh Mixed Meat; AMM=Autoclaved Mixed Meat; SMM=Steamed Mixed Meat; AMP=Autoclaved Mixed Meat Powder; SMP=Steamed Mixed Meat Powder.
Conclusion
This study provides insights into the effects of heat treatment on mackerel tuna's nutritional profile, molecular weight distribution, and allergenicity, highlighting its potential as a hypoallergenic food source. The findings indicate that both pressure heating and steaming significantly reduce moisture content while increasing protein levels in fish powder. Moreover, pressure heating effectively eliminates allergenic protein bands with molecular weights of 10-11 kDa, whereas steaming increases band intensity. Analysis of allergen levels in mackerel tuna showed that pressure heating significantly reduced allergenicity in white, red, and mixed meat with a greater reduction than steaming. Additionally, the drying and grinding process further enhanced allergen reduction, with white meat showing the greatest effect, followed by red and mixed meat. Among the meat types, white meat exhibited the highest parvalbumin content, followed by red and mixed meats. Considering chemical properties, lower allergenicity, and better economic value, mixed meat (ratio of three parts white meat to one part red meat) is recommended for fish powder production over the other meat types. The study did not include sensory evaluation or consumer acceptance testing, which are essential for assessing market feasibility. Furthermore, although economic and sustainability aspects were considered, a detailed cost-benefit and environmental impact analysis was not conducted; future research can focus on these parameters.
Author contributions
Z.K. conducted the experiment, analyzed data, interpreted statistical analysis, and wrote the main draft of the manuscript; N.S.P. and N.W. supervised the research and critically revised the article for important intellectual content; F.K. participated in editing of the original draft; All authors read and approved the final manuscript.
Acknowledgments
The authors are grateful to the Head of the Department of Food Science and Technology, Faculty of Agricultural Engineering and Technology, IPB University, Bogor, Indonesia, for the permission to use the Departmental laboratory.
Conflicts of interest
The authors declared that there is no conflict of interest.
Funding
This research was partially funded by Lembaga Pengelola Dana Pendidikan (LPDP), Ministry of Finance of the Republic of Indonesia and partially facilitated through research projects supported by the Directorate General of Higher Education, Research, and Technology, Ministry of Education, Culture, Research, and Technology.
Ethical Consideration
Not applicable.
Reference
Abbott M., Hayward S., Ross W., Godefroy S.B., Ulberth F., Van Hengel A.J., Roberts J., Akiyama H., Popping B., Yeung J.M., Wehling P., Taylor S.L., et al. (2010). Validation procedures for quantitative food allergen ELISA methods: community guidance and best practices. Journal of AOAC International. 93: 442-450. [DOI: 10.1093/jaoac/93.2.442]
Adawyah R., Khotiffah S.K., Wahyudinur, Puspitasari F. (2020). Effect of different cooking time on protein and fat content, amino acid and fatty acid profile of three spot gourami (Trichogaster trichopterus) fish flour. Jurnal Pengolahan Hasil Perikanan Indonesia. 23: 286-294. [Indonesian with English abstract]
Akhtara N., Borah A. (2022). Influence of microwave and traditional smoked drying method on the protein and moisture content of Puntius sophore. International Journal of Biochemistry Research and Review. 31: 10-16. [DOI: 10.9734/IJBCRR/2022/v31i9777]
Albrecht-Ruiz M., Salas-Maldonado A. (2015). Chemical composition of light and dark muscle of peruvian anchovy (Engraulis ringens) and its seasonal variation. Journal of Aquatic Food Product Technology. 24: 191-196. [DOI: 10.1080/ 10498850. 2012.762705]
Al Sailawi H.A., Misnan R., Yadzir Z.H.M., Abdullah N., Bakhtiar F., Arip M., Mudhafar M., Ateshan H.M. (2020). Effects of different salting and drying methods on allergenicity of purple mud crab (Scylla tranquebarica). Indian Journal of Ecology. 47: 1173-1179.
AOAC International. (1995). Official methods of analysis of AOAC international. 16th edition. Journal of AOAC International. Arlington, USA. URL: http://lib3.dss.go.th/fulltext/scan_ ebook/aoac_1995_v78_n3.pdf. Accessed 1 May 2023.
Ariva A.N., Widyasanti A., Nurjanah S. (2020). The effect of drying temperature to the quality of cascara tea from arabica pulp (Coffea arabica). Jurnal Teknologi dan Industri Pertanian Indonesia. 12: 21-28. [DOI: 10.17969/jtipi.v12i1.15744]. [Indonesian with English abstract]
Bastías J.M., Balladares P., Acuña S., Quevedo R., Muñoz O. (2017). Determining the effect of different cooking methods on the nutritional composition of salmon (Salmo salar) and chilean jack mackerel (Trachurus murphyi) fillets. Plos One. 12: e0180993. [DOI: 10.1371/journal.pone.0180993]
Chen Y.-T., Hsieh Y.-H.P. (2021). Development and characterization of monoclonal antibodies for the detection of fish protein. Foods. 10: 2360. [DOI: 10.3390/foods10102360]
Dasanayaka B.P., Li Z., Pramod S.N., Chen Y., Khan M.U., Lin H. (2020). A review on food processing and preparation methods for altering fish allergenicity. Critical Reviews in Food Science and Nutrition. 62: 1951-1970. [DOI: 10.1080/ 10408398.2020.1848791]
Dasanayaka B.P., Wang H., Li Z., Yu M., Ahmed A.M.M., Zhang Z., Lin H., Wang X. (2022). Evaluating the effects of processing on antigenicity and immunochemical detectability of fish proteins by ELISA. Journal of Food Composition and Analysis. 112: 104690. [DOI: 10.1016/j.jfca.2022.104690]
De Jongh H.H.J., Jimenez M.D.L.R., Baumert J.L., Taylor S.L., Koppelman S.J. (2015). Electrophoretic behavior in relation to the structural integrity of codfish parvalbumin upon heat treatment. Journal of Agricultural and Food Chemistry. 63: 4683-4689. [DOI: 10.1021/jf505990h]
Kamath S.D., Bublin M., Kitamura K., Matsui T., Ito K., Lopata A.L. (2023). Cross-reactive epitopes and their role in food allergy. Journal of Allergy and Clinical Immunology. 151: 1178-1190. [DOI: 10.1016/j.jaci.2022.12.827]
Kannaiyan S.K., Bagthasingh C., Vetri V., Aran S.S., Venkatachalam K. (2019). Nutritional, textural and quality attributes of white and dark muscles of little tuna (Euthynnus affinis). Indian Journal of Geo Marine Sciences. 48: 205-211.
Khasanah M.M., Ujianti R.M.D., Nurdyansyah F., Ferdiansyah D.M.K. (2020). Characteristics of milk fish (Chanos chanos) crackers with varied types of fish flour processing and starch. Jurnal Pascapanen Dan Bioteknologi Kelautan Dan Perikanan. 15: 143-149. [DOI: 10.15578/jpbkp.v15i2.634]. [Indonesian with English abstract]
Kilic A. (2022). Experimental identification of thermal steam pressure sensitivity of ω-3 fatty acids in fish. Journal of Culinary Science and Technology. 22: 386-401. [DOI: 10.1080/15428052.2022. 2051661]
Kobayashi Y., Yang T., Yu C.-T., Ume C., Kubota H., Shimakura K., Shiomi K., Hamada-Sato N. (2016). Quantification of major allergen parvalbumin in 22 species of fish by SDS-PAGE. Food Chemistry. 194: 345-353. [DOI: 10.1016/j.foodchem.2015. 08.037]
Kong X., Qiu X., Li P., Li Y., Zhang Y., Guo X., Kong F. (2024). Enhancement of nutrient bioaccessibility and functional property of chicken bone powder through steam explosion. Journal of Agriculture and Food Research. 15: 100941. [DOI: 10.1016/j.jafr.2023.100941]
Kubota H., Kobayashi A., Kobayashi Y., Shiomi K., Hamada-Sato N. (2016). Reduction in IgE reactivity of pacific mackerel parvalbumin by heat treatment. Food Chemistry. 206: 78-84. [DOI: 10.1016/j.foodchem.2016.03.043]
Kuniyoshi M.L.G., Da Silva-Gomes R.N., Zanella B.T.T., Dos Santos L.D., Dal-Pai-Silva M. (2019). Anabolism, catabolism and proteomic analysis in the slow-twitch muscle of pacu (Piaractus mesopotamicus) submitted to prolonged fasting followed by refeeding. Aquaculture Research. 51: 1101-1112. [DOI: 10.1111/are.14456]
Laemmli U.K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 227: 680-685.
Lakmini K.P.C., Gonapinuwala S.T., Senarath H.P.S., Fernando C.A.N., Wijesekara I., De Croos M.D.S.T. (2022). Effect of autoclaving as a pre-treatment in the wet reduction process for extracting fish oil from yellowfin tuna heads. Sri Lanka Journal of Aquatic Sciences. 27: 43-61. [DOI: 10.4038/sljas.v27i1.7596]
Laly S.J., Sankar T.V., Panda S.K. (2022). Effect of pressure cooking alone and in combination with other treatments on shrimp allergic protein, tropomyosin. Journal of Food Science and Technology. 59: 1193-1201. [DOI: 10.1007/s13197-021-05124-2]
Li Q., Zhang L., Lu H., Song S., Luo Y. (2017). Comparison of postmortem changes in ATP-related compounds, protein degradation and endogenous enzyme activity of white muscle and dark muscle from common carp (Cyprinus carpio) stored at 4 °C. LWT. 78: 317-324. [DOI: 10.1016/j.lwt.2016.12.035]
Li Z., Luo Y., Jiang M. (2016). Effects of heat treatment on the antigenicity and allergenicity of grass carp muscles. Journal of Aquatic Food Product Technology. 25: 350-357. [DOI: 10.1080/10498850.2013.851758]
Mardlijah S., Pane A.R.P., Fauzi M., Yusuf H.N., Widiyastuti H., Herlisman, Zamroni A., Noegroho T., Hufiadi, Wagiyo K. (2022). The fishing grounds and the exploitation status of Kawakawa (Euthynnus affinis) in Java sea, Indonesia. HAYATI Journal of Biosciences. 29: 255-265. [DOI: 10.4308/ hjb.29.2.255-265]
Mastrorilli C., Arasi S., Barni S., Caimmi D., Chiera F., Comberiati P., Dinardo G., Giannetti A., Gismondi M., Gracci S., Paravati F., Pelosi U., et al. (2023). IgE-mediated and non-IgE-mediated fish allergy in pediatric age: a holistic approach—a consensus by diagnostic commission of the Italian society of pediatric allergy and immunology. Medicina. 59: 1651. [DOI: 10.3390/ medicina59091651]
Maulidah R., Istyadji M., Sauqina. (2022). Pengaruh lama pemasakan yang berbeda terhadap kadar protein dan lemak tepung ikan papuyu (Anabas testudineus). JUSTER: Jurnal Sains dan Terapan. 1: 171-179. [DOI: 10.57218/juster.v1i3.370]. [Indonesian with English abstract]
Nguyen H.T., Hilmarsdóttir G.S., Tómasson T., Arason S., Gudjónsdóttir M. (2022). Changes in protein and non-protein nitrogen compounds during fishmeal processing—identification of unoptimized processing steps. Processes. 10: 621. [DOI: 10.3390/pr10040621]
Nugraha R., Ruethers T., Johnston E.B., Rolland J.M., O’hehir R.E., Kamath S.D., Lopata A.L. (2021). Effects of extraction buffer on the solubility and immunoreactivity of the pacific oyster allergens. Foods. 10: 16. [DOI: 10.3390/foods10020409]
Ochiai Y., Ozawa H. (2020). Biochemical and physicochemical characteristics of the major muscle proteins from fish and shellfish. Fisheries Science. 86: 729-740. [DOI: 10.1007/s12562-020-01444-y]
Palupi N.S., Indrastuti N.A., Wulandari N. (2021). Indonesian traditional salted fish: the alteration its allergenicity during processing. Journal of Aquatic Food Product Technology. 30: 353-363. [DOI: 10.1080/10498850.2021.1882632]
Pramestia S.P., Riyanto B., Trilaksani W. (2015). Fish oil microencapsulation as omega-3 fatty acids fortification material for cream of crab soup. Jurnal Pengolahan Hasil Perikanan Indonesia. 18: 162-176. [DOI: 10.17844/jphpi.2015.18.2.162]. [Indonesian with English abstract]
Prester L. (2016). Seafood allergy, toxicity, and intolerance: a review. Journal of the American College of Nutrition. 35: 271-283. [DOI: 10.1080/07315724.2015.1014120]
Rittisak S., Charoen R., Savedboworn W. (2022). Broken riceberry (br) powder production using a double drum dryer and its utilization in the development of instant beverages. Processes. 10: 341. [DOI: 10.3390/pr10020341]
Ruethers T., Taki A.C., Johnston E.B., Nugraha R., Le T.T.K., Kalic T., McLean T.R., Kamath S.D., Lopata A.L. (2018). Seafood allergy: a comprehensive review of fish and shellfish allergens. Molecular Immunology. 100: 28-57. [DOI: 10.1016/j. molimm.2018.04.008]
Schrama D., Raposo De Magalhães C., Cerqueira M., Carrilho R., Revets D., Kuehn A., Engrola S., Rodrigues P.M. (2022). Fish processing and digestion affect parvalbumins detectability in gilthead seabream and European seabass. Animals. 12: 3022. [DOI: 10.3390/ani12213022]
Soegiarto G., Abdullah M.S., Damayanti L.A., Suseno A., Effendi C. (2019). The prevalence of allergic diseases in school children of metropolitan city in Indonesia shows a similar pattern to that of developed countries. Asia Pacific Allergy. 9: e17. [DOI: 10.5415/apallergy.2019.9.e17]
Stevens J.R., Newton R.W., Tlusty M., Little D.C. (2018). The rise of aquaculture by-products: increasing food production, value, and sustainability through strategic utilisation. Marine Policy. 90: 115-124. [DOI: 10.1016/j.marpol.2017.12.027]
Strasburg G., Xiong Y.L., Chiang W. (2017). Physiology and chemistry of edible muscle tissues. In: Damodaran S., Parkin K.L., Fennema O.R. (Editors). Fennema’s food chemistry. 5th edition. CRC Press, Boca Raton. pp: 923-974. [DOI: 10.1201/9781315372914]
Tedner S.G., Asarnoj A., Thulin H., Westman M., Konradsen J.R., Nilsson C. (2022). Food allergy and hypersensitivity reactions in children and adults—a review. Journal of Internal Medicine. 291: 283-302. [DOI: 10.1111/joim.13422]
Tiwari M., Barooah M.S., Sharma P., Bordoloi P.L., Hussain I.A., Ahmed A.M. (2021). Physico–chemical characteristics of fish flour prepared from locally available small indigenous fish species of Assam. Journal of Food Processing and Preservation. 45: e15704. [DOI: 10.1111/jfpp.15704]
Tontul I., Topuz A. (2017). Spray-drying of fruit and vegetable juices: Effect of drying conditions on the product yield and physical properties. Trends in Food Science and Technology. 63: 91-102. [DOI: 10.1016/j.tifs.2017.03.009]
Tsai C.-L., Perng K., Hou Y.-C., Shen C.-J., Chen I.-N., Chen Y.-T. (2023). Effect of species, muscle location, food processing and refrigerated storage on the fish allergens, tropomyosin and parvalbumin. Food Chemistry. 402: 134479. [DOI: 10.1016/j.foodchem.2022.134479]
Vanga S.K., Singh A., Raghavan V. (2017). Review of conventional and novel food processing methods on food allergens. Critical Reviews in Food Science and Nutrition. 57: 2077-2094. [DOI: 10.1080/10408398.2015.1045965]
World Allergy Organization (WAO). (2013). WAO white book on allergy: update 2013, executive summary. WAO, USA. URL: https://allergypaais.org/wp-content/themes/twentytwentyone/ pdf/ExecSummary-2013-v6-hires.pdf.
Wulan N.L.N., Palupi N.S., Kusnandar F., Wulandari N. (2024). Autoclave process contribution to the production of black tiger shrimp powder (Penaeus monodon) and hypoallergenic puffing snacks. Jurnal Pengolahan Hasil Perikanan Indonesia. 27: 142-158. [DOI: 10.17844/jphpi.v27i2.50620]. [Indonesian with English abstract]
Zhou F., He S., Sun H., Wang Y., Zhang Y. (2021). Advances in epitope mapping technologies for food protein allergens: a review. Trends in Food Science and Technology. 107: 226-239. [DOI: 10.1016/j.tifs.2020.10.035]
This study provides insights into the effects of heat treatment on mackerel tuna's nutritional profile, molecular weight distribution, and allergenicity, highlighting its potential as a hypoallergenic food source. The findings indicate that both pressure heating and steaming significantly reduce moisture content while increasing protein levels in fish powder. Moreover, pressure heating effectively eliminates allergenic protein bands with molecular weights of 10-11 kDa, whereas steaming increases band intensity. Analysis of allergen levels in mackerel tuna showed that pressure heating significantly reduced allergenicity in white, red, and mixed meat with a greater reduction than steaming. Additionally, the drying and grinding process further enhanced allergen reduction, with white meat showing the greatest effect, followed by red and mixed meat. Among the meat types, white meat exhibited the highest parvalbumin content, followed by red and mixed meats. Considering chemical properties, lower allergenicity, and better economic value, mixed meat (ratio of three parts white meat to one part red meat) is recommended for fish powder production over the other meat types. The study did not include sensory evaluation or consumer acceptance testing, which are essential for assessing market feasibility. Furthermore, although economic and sustainability aspects were considered, a detailed cost-benefit and environmental impact analysis was not conducted; future research can focus on these parameters.
Author contributions
Z.K. conducted the experiment, analyzed data, interpreted statistical analysis, and wrote the main draft of the manuscript; N.S.P. and N.W. supervised the research and critically revised the article for important intellectual content; F.K. participated in editing of the original draft; All authors read and approved the final manuscript.
Acknowledgments
The authors are grateful to the Head of the Department of Food Science and Technology, Faculty of Agricultural Engineering and Technology, IPB University, Bogor, Indonesia, for the permission to use the Departmental laboratory.
Conflicts of interest
The authors declared that there is no conflict of interest.
Funding
This research was partially funded by Lembaga Pengelola Dana Pendidikan (LPDP), Ministry of Finance of the Republic of Indonesia and partially facilitated through research projects supported by the Directorate General of Higher Education, Research, and Technology, Ministry of Education, Culture, Research, and Technology.
Ethical Consideration
Not applicable.
Reference
Abbott M., Hayward S., Ross W., Godefroy S.B., Ulberth F., Van Hengel A.J., Roberts J., Akiyama H., Popping B., Yeung J.M., Wehling P., Taylor S.L., et al. (2010). Validation procedures for quantitative food allergen ELISA methods: community guidance and best practices. Journal of AOAC International. 93: 442-450. [DOI: 10.1093/jaoac/93.2.442]
Adawyah R., Khotiffah S.K., Wahyudinur, Puspitasari F. (2020). Effect of different cooking time on protein and fat content, amino acid and fatty acid profile of three spot gourami (Trichogaster trichopterus) fish flour. Jurnal Pengolahan Hasil Perikanan Indonesia. 23: 286-294. [Indonesian with English abstract]
Akhtara N., Borah A. (2022). Influence of microwave and traditional smoked drying method on the protein and moisture content of Puntius sophore. International Journal of Biochemistry Research and Review. 31: 10-16. [DOI: 10.9734/IJBCRR/2022/v31i9777]
Albrecht-Ruiz M., Salas-Maldonado A. (2015). Chemical composition of light and dark muscle of peruvian anchovy (Engraulis ringens) and its seasonal variation. Journal of Aquatic Food Product Technology. 24: 191-196. [DOI: 10.1080/ 10498850. 2012.762705]
Al Sailawi H.A., Misnan R., Yadzir Z.H.M., Abdullah N., Bakhtiar F., Arip M., Mudhafar M., Ateshan H.M. (2020). Effects of different salting and drying methods on allergenicity of purple mud crab (Scylla tranquebarica). Indian Journal of Ecology. 47: 1173-1179.
AOAC International. (1995). Official methods of analysis of AOAC international. 16th edition. Journal of AOAC International. Arlington, USA. URL: http://lib3.dss.go.th/fulltext/scan_ ebook/aoac_1995_v78_n3.pdf. Accessed 1 May 2023.
Ariva A.N., Widyasanti A., Nurjanah S. (2020). The effect of drying temperature to the quality of cascara tea from arabica pulp (Coffea arabica). Jurnal Teknologi dan Industri Pertanian Indonesia. 12: 21-28. [DOI: 10.17969/jtipi.v12i1.15744]. [Indonesian with English abstract]
Bastías J.M., Balladares P., Acuña S., Quevedo R., Muñoz O. (2017). Determining the effect of different cooking methods on the nutritional composition of salmon (Salmo salar) and chilean jack mackerel (Trachurus murphyi) fillets. Plos One. 12: e0180993. [DOI: 10.1371/journal.pone.0180993]
Chen Y.-T., Hsieh Y.-H.P. (2021). Development and characterization of monoclonal antibodies for the detection of fish protein. Foods. 10: 2360. [DOI: 10.3390/foods10102360]
Dasanayaka B.P., Li Z., Pramod S.N., Chen Y., Khan M.U., Lin H. (2020). A review on food processing and preparation methods for altering fish allergenicity. Critical Reviews in Food Science and Nutrition. 62: 1951-1970. [DOI: 10.1080/ 10408398.2020.1848791]
Dasanayaka B.P., Wang H., Li Z., Yu M., Ahmed A.M.M., Zhang Z., Lin H., Wang X. (2022). Evaluating the effects of processing on antigenicity and immunochemical detectability of fish proteins by ELISA. Journal of Food Composition and Analysis. 112: 104690. [DOI: 10.1016/j.jfca.2022.104690]
De Jongh H.H.J., Jimenez M.D.L.R., Baumert J.L., Taylor S.L., Koppelman S.J. (2015). Electrophoretic behavior in relation to the structural integrity of codfish parvalbumin upon heat treatment. Journal of Agricultural and Food Chemistry. 63: 4683-4689. [DOI: 10.1021/jf505990h]
Kamath S.D., Bublin M., Kitamura K., Matsui T., Ito K., Lopata A.L. (2023). Cross-reactive epitopes and their role in food allergy. Journal of Allergy and Clinical Immunology. 151: 1178-1190. [DOI: 10.1016/j.jaci.2022.12.827]
Kannaiyan S.K., Bagthasingh C., Vetri V., Aran S.S., Venkatachalam K. (2019). Nutritional, textural and quality attributes of white and dark muscles of little tuna (Euthynnus affinis). Indian Journal of Geo Marine Sciences. 48: 205-211.
Khasanah M.M., Ujianti R.M.D., Nurdyansyah F., Ferdiansyah D.M.K. (2020). Characteristics of milk fish (Chanos chanos) crackers with varied types of fish flour processing and starch. Jurnal Pascapanen Dan Bioteknologi Kelautan Dan Perikanan. 15: 143-149. [DOI: 10.15578/jpbkp.v15i2.634]. [Indonesian with English abstract]
Kilic A. (2022). Experimental identification of thermal steam pressure sensitivity of ω-3 fatty acids in fish. Journal of Culinary Science and Technology. 22: 386-401. [DOI: 10.1080/15428052.2022. 2051661]
Kobayashi Y., Yang T., Yu C.-T., Ume C., Kubota H., Shimakura K., Shiomi K., Hamada-Sato N. (2016). Quantification of major allergen parvalbumin in 22 species of fish by SDS-PAGE. Food Chemistry. 194: 345-353. [DOI: 10.1016/j.foodchem.2015. 08.037]
Kong X., Qiu X., Li P., Li Y., Zhang Y., Guo X., Kong F. (2024). Enhancement of nutrient bioaccessibility and functional property of chicken bone powder through steam explosion. Journal of Agriculture and Food Research. 15: 100941. [DOI: 10.1016/j.jafr.2023.100941]
Kubota H., Kobayashi A., Kobayashi Y., Shiomi K., Hamada-Sato N. (2016). Reduction in IgE reactivity of pacific mackerel parvalbumin by heat treatment. Food Chemistry. 206: 78-84. [DOI: 10.1016/j.foodchem.2016.03.043]
Kuniyoshi M.L.G., Da Silva-Gomes R.N., Zanella B.T.T., Dos Santos L.D., Dal-Pai-Silva M. (2019). Anabolism, catabolism and proteomic analysis in the slow-twitch muscle of pacu (Piaractus mesopotamicus) submitted to prolonged fasting followed by refeeding. Aquaculture Research. 51: 1101-1112. [DOI: 10.1111/are.14456]
Laemmli U.K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 227: 680-685.
Lakmini K.P.C., Gonapinuwala S.T., Senarath H.P.S., Fernando C.A.N., Wijesekara I., De Croos M.D.S.T. (2022). Effect of autoclaving as a pre-treatment in the wet reduction process for extracting fish oil from yellowfin tuna heads. Sri Lanka Journal of Aquatic Sciences. 27: 43-61. [DOI: 10.4038/sljas.v27i1.7596]
Laly S.J., Sankar T.V., Panda S.K. (2022). Effect of pressure cooking alone and in combination with other treatments on shrimp allergic protein, tropomyosin. Journal of Food Science and Technology. 59: 1193-1201. [DOI: 10.1007/s13197-021-05124-2]
Li Q., Zhang L., Lu H., Song S., Luo Y. (2017). Comparison of postmortem changes in ATP-related compounds, protein degradation and endogenous enzyme activity of white muscle and dark muscle from common carp (Cyprinus carpio) stored at 4 °C. LWT. 78: 317-324. [DOI: 10.1016/j.lwt.2016.12.035]
Li Z., Luo Y., Jiang M. (2016). Effects of heat treatment on the antigenicity and allergenicity of grass carp muscles. Journal of Aquatic Food Product Technology. 25: 350-357. [DOI: 10.1080/10498850.2013.851758]
Mardlijah S., Pane A.R.P., Fauzi M., Yusuf H.N., Widiyastuti H., Herlisman, Zamroni A., Noegroho T., Hufiadi, Wagiyo K. (2022). The fishing grounds and the exploitation status of Kawakawa (Euthynnus affinis) in Java sea, Indonesia. HAYATI Journal of Biosciences. 29: 255-265. [DOI: 10.4308/ hjb.29.2.255-265]
Mastrorilli C., Arasi S., Barni S., Caimmi D., Chiera F., Comberiati P., Dinardo G., Giannetti A., Gismondi M., Gracci S., Paravati F., Pelosi U., et al. (2023). IgE-mediated and non-IgE-mediated fish allergy in pediatric age: a holistic approach—a consensus by diagnostic commission of the Italian society of pediatric allergy and immunology. Medicina. 59: 1651. [DOI: 10.3390/ medicina59091651]
Maulidah R., Istyadji M., Sauqina. (2022). Pengaruh lama pemasakan yang berbeda terhadap kadar protein dan lemak tepung ikan papuyu (Anabas testudineus). JUSTER: Jurnal Sains dan Terapan. 1: 171-179. [DOI: 10.57218/juster.v1i3.370]. [Indonesian with English abstract]
Nguyen H.T., Hilmarsdóttir G.S., Tómasson T., Arason S., Gudjónsdóttir M. (2022). Changes in protein and non-protein nitrogen compounds during fishmeal processing—identification of unoptimized processing steps. Processes. 10: 621. [DOI: 10.3390/pr10040621]
Nugraha R., Ruethers T., Johnston E.B., Rolland J.M., O’hehir R.E., Kamath S.D., Lopata A.L. (2021). Effects of extraction buffer on the solubility and immunoreactivity of the pacific oyster allergens. Foods. 10: 16. [DOI: 10.3390/foods10020409]
Ochiai Y., Ozawa H. (2020). Biochemical and physicochemical characteristics of the major muscle proteins from fish and shellfish. Fisheries Science. 86: 729-740. [DOI: 10.1007/s12562-020-01444-y]
Palupi N.S., Indrastuti N.A., Wulandari N. (2021). Indonesian traditional salted fish: the alteration its allergenicity during processing. Journal of Aquatic Food Product Technology. 30: 353-363. [DOI: 10.1080/10498850.2021.1882632]
Pramestia S.P., Riyanto B., Trilaksani W. (2015). Fish oil microencapsulation as omega-3 fatty acids fortification material for cream of crab soup. Jurnal Pengolahan Hasil Perikanan Indonesia. 18: 162-176. [DOI: 10.17844/jphpi.2015.18.2.162]. [Indonesian with English abstract]
Prester L. (2016). Seafood allergy, toxicity, and intolerance: a review. Journal of the American College of Nutrition. 35: 271-283. [DOI: 10.1080/07315724.2015.1014120]
Rittisak S., Charoen R., Savedboworn W. (2022). Broken riceberry (br) powder production using a double drum dryer and its utilization in the development of instant beverages. Processes. 10: 341. [DOI: 10.3390/pr10020341]
Ruethers T., Taki A.C., Johnston E.B., Nugraha R., Le T.T.K., Kalic T., McLean T.R., Kamath S.D., Lopata A.L. (2018). Seafood allergy: a comprehensive review of fish and shellfish allergens. Molecular Immunology. 100: 28-57. [DOI: 10.1016/j. molimm.2018.04.008]
Schrama D., Raposo De Magalhães C., Cerqueira M., Carrilho R., Revets D., Kuehn A., Engrola S., Rodrigues P.M. (2022). Fish processing and digestion affect parvalbumins detectability in gilthead seabream and European seabass. Animals. 12: 3022. [DOI: 10.3390/ani12213022]
Soegiarto G., Abdullah M.S., Damayanti L.A., Suseno A., Effendi C. (2019). The prevalence of allergic diseases in school children of metropolitan city in Indonesia shows a similar pattern to that of developed countries. Asia Pacific Allergy. 9: e17. [DOI: 10.5415/apallergy.2019.9.e17]
Stevens J.R., Newton R.W., Tlusty M., Little D.C. (2018). The rise of aquaculture by-products: increasing food production, value, and sustainability through strategic utilisation. Marine Policy. 90: 115-124. [DOI: 10.1016/j.marpol.2017.12.027]
Strasburg G., Xiong Y.L., Chiang W. (2017). Physiology and chemistry of edible muscle tissues. In: Damodaran S., Parkin K.L., Fennema O.R. (Editors). Fennema’s food chemistry. 5th edition. CRC Press, Boca Raton. pp: 923-974. [DOI: 10.1201/9781315372914]
Tedner S.G., Asarnoj A., Thulin H., Westman M., Konradsen J.R., Nilsson C. (2022). Food allergy and hypersensitivity reactions in children and adults—a review. Journal of Internal Medicine. 291: 283-302. [DOI: 10.1111/joim.13422]
Tiwari M., Barooah M.S., Sharma P., Bordoloi P.L., Hussain I.A., Ahmed A.M. (2021). Physico–chemical characteristics of fish flour prepared from locally available small indigenous fish species of Assam. Journal of Food Processing and Preservation. 45: e15704. [DOI: 10.1111/jfpp.15704]
Tontul I., Topuz A. (2017). Spray-drying of fruit and vegetable juices: Effect of drying conditions on the product yield and physical properties. Trends in Food Science and Technology. 63: 91-102. [DOI: 10.1016/j.tifs.2017.03.009]
Tsai C.-L., Perng K., Hou Y.-C., Shen C.-J., Chen I.-N., Chen Y.-T. (2023). Effect of species, muscle location, food processing and refrigerated storage on the fish allergens, tropomyosin and parvalbumin. Food Chemistry. 402: 134479. [DOI: 10.1016/j.foodchem.2022.134479]
Vanga S.K., Singh A., Raghavan V. (2017). Review of conventional and novel food processing methods on food allergens. Critical Reviews in Food Science and Nutrition. 57: 2077-2094. [DOI: 10.1080/10408398.2015.1045965]
World Allergy Organization (WAO). (2013). WAO white book on allergy: update 2013, executive summary. WAO, USA. URL: https://allergypaais.org/wp-content/themes/twentytwentyone/ pdf/ExecSummary-2013-v6-hires.pdf.
Wulan N.L.N., Palupi N.S., Kusnandar F., Wulandari N. (2024). Autoclave process contribution to the production of black tiger shrimp powder (Penaeus monodon) and hypoallergenic puffing snacks. Jurnal Pengolahan Hasil Perikanan Indonesia. 27: 142-158. [DOI: 10.17844/jphpi.v27i2.50620]. [Indonesian with English abstract]
Zhou F., He S., Sun H., Wang Y., Zhang Y. (2021). Advances in epitope mapping technologies for food protein allergens: a review. Trends in Food Science and Technology. 107: 226-239. [DOI: 10.1016/j.tifs.2020.10.035]
[*] Corresponding author (N. Sri Palupi)
* E-mail: hnpalupi@apps.ipb.ac.id
ORCID ID: https://orcid.org/0000-0003-1708-002X
* E-mail: hnpalupi@apps.ipb.ac.id
ORCID ID: https://orcid.org/0000-0003-1708-002X
Type of Study: Original article |
Subject:
Special
Received: 24/11/09 | Accepted: 25/05/21 | Published: 25/06/22
Received: 24/11/09 | Accepted: 25/05/21 | Published: 25/06/22
References
1. Abbott M., Hayward S., Ross W., Godefroy S.B., Ulberth F., Van Hengel A.J., Roberts J., Akiyama H., Popping B., Yeung J.M., Wehling P., Taylor S.L., et al. (2010). Validation procedures for quantitative food allergen ELISA methods: community guidance and best practices. Journal of AOAC International. 93: 442-450. [DOI: 10.1093/jaoac/93.2.442] [DOI:10.1093/jaoac/93.2.442] [PMID]
2. Adawyah R., Khotiffah S.K., Wahyudinur, Puspitasari F. (2020). Effect of different cooking time on protein and fat content, amino acid and fatty acid profile of three spot gourami (Trichogaster trichopterus) fish flour. Jurnal Pengolahan Hasil Perikanan Indonesia. 23: 286-294. [Indonesian with English abstract] [DOI:10.17844/jphpi.v23i2.32339]
3. Akhtara N., Borah A. (2022). Influence of microwave and traditional smoked drying method on the protein and moisture content of Puntius sophore. International Journal of Biochemistry Research and Review. 31: 10-16. [DOI: 10.9734/IJBCRR/2022/v31i9777] [DOI:10.9734/ijbcrr/2022/v31i9777]
4. Albrecht-Ruiz M., Salas-Maldonado A. (2015). Chemical composition of light and dark muscle of peruvian anchovy (Engraulis ringens) and its seasonal variation. Journal of Aquatic Food Product Technology. 24: 191-196. [DOI: 10.1080/ 10498850. 2012.762705] [DOI:10.1080/10498850.2012.762705]
5. Al Sailawi H.A., Misnan R., Yadzir Z.H.M., Abdullah N., Bakhtiar F., Arip M., Mudhafar M., Ateshan H.M. (2020). Effects of different salting and drying methods on allergenicity of purple mud crab (Scylla tranquebarica). Indian Journal of Ecology. 47: 1173-1179.
6. AOAC International. (1995). Official methods of analysis of AOAC international. 16th edition. Journal of AOAC International. Arlington, USA. URL: http://lib3.dss.go.th/fulltext/scan_ ebook/aoac_1995_v78_n3.pdf. Accessed 1 May 2023.
7. Ariva A.N., Widyasanti A., Nurjanah S. (2020). The effect of drying temperature to the quality of cascara tea from arabica pulp (Coffea arabica). Jurnal Teknologi dan Industri Pertanian Indonesia. 12: 21-28. [DOI: 10.17969/jtipi.v12i1.15744]. [Indonesian with English abstract] [DOI:10.17969/jtipi.v12i1.15744]
8. Bastías J.M., Balladares P., Acuña S., Quevedo R., Muñoz O. (2017). Determining the effect of different cooking methods on the nutritional composition of salmon (Salmo salar) and chilean jack mackerel (Trachurus murphyi) fillets. Plos One. 12: e0180993. [DOI: 10.1371/journal.pone.0180993] [DOI:10.1371/journal.pone.0180993] [PMID] [PMCID]
9. Chen Y.-T., Hsieh Y.-H.P. (2021). Development and characterization of monoclonal antibodies for the detection of fish protein. Foods. 10: 2360. [DOI: 10.3390/foods10102360] [DOI:10.3390/foods10102360] [PMID] [PMCID]
10. Dasanayaka B.P., Li Z., Pramod S.N., Chen Y., Khan M.U., Lin H. (2020). A review on food processing and preparation methods for altering fish allergenicity. Critical Reviews in Food Science and Nutrition. 62: 1951-1970. [DOI: 10.1080/ 10408398.2020.1848791] [DOI:10.1080/10408398.2020.1848791] [PMID]
11. Dasanayaka B.P., Wang H., Li Z., Yu M., Ahmed A.M.M., Zhang Z., Lin H., Wang X. (2022). Evaluating the effects of processing on antigenicity and immunochemical detectability of fish proteins by ELISA. Journal of Food Composition and Analysis. 112: 104690. [DOI: 10.1016/j.jfca.2022.104690] [DOI:10.1016/j.jfca.2022.104690]
12. De Jongh H.H.J., Jimenez M.D.L.R., Baumert J.L., Taylor S.L., Koppelman S.J. (2015). Electrophoretic behavior in relation to the structural integrity of codfish parvalbumin upon heat treatment. Journal of Agricultural and Food Chemistry. 63: 4683-4689. [DOI: 10.1021/jf505990h] [DOI:10.1021/jf505990h] [PMID]
13. Kamath S.D., Bublin M., Kitamura K., Matsui T., Ito K., Lopata A.L. (2023). Cross-reactive epitopes and their role in food allergy. Journal of Allergy and Clinical Immunology. 151: 1178-1190. [DOI: 10.1016/j.jaci.2022.12.827] [DOI:10.1016/j.jaci.2022.12.827] [PMID]
14. Kannaiyan S.K., Bagthasingh C., Vetri V., Aran S.S., Venkatachalam K. (2019). Nutritional, textural and quality attributes of white and dark muscles of little tuna (Euthynnus affinis). Indian Journal of Geo Marine Sciences. 48: 205-211.
15. Khasanah M.M., Ujianti R.M.D., Nurdyansyah F., Ferdiansyah D.M.K. (2020). Characteristics of milk fish (Chanos chanos) crackers with varied types of fish flour processing and starch. Jurnal Pascapanen Dan Bioteknologi Kelautan Dan Perikanan. 15: 143-149. [DOI: 10.15578/jpbkp.v15i2.634]. [Indonesian with English abstract] [DOI:10.15578/jpbkp.v15i2.634]
16. Kilic A. (2022). Experimental identification of thermal steam pressure sensitivity of ω-3 fatty acids in fish. Journal of Culinary Science and Technology. 22: 386-401. [DOI: 10.1080/15428052.2022. 2051661] [DOI:10.1080/15428052.2022.2051661]
17. Kobayashi Y., Yang T., Yu C.-T., Ume C., Kubota H., Shimakura K., Shiomi K., Hamada-Sato N. (2016). Quantification of major allergen parvalbumin in 22 species of fish by SDS-PAGE. Food Chemistry. 194: 345-353. [DOI: 10.1016/j.foodchem.2015. 08.037] [DOI:10.1016/j.foodchem.2015.08.037] [PMID]
18. Kong X., Qiu X., Li P., Li Y., Zhang Y., Guo X., Kong F. (2024). Enhancement of nutrient bioaccessibility and functional property of chicken bone powder through steam explosion. Journal of Agriculture and Food Research. 15: 100941. [DOI: 10.1016/j.jafr.2023.100941] [DOI:10.1016/j.jafr.2023.100941]
19. Kubota H., Kobayashi A., Kobayashi Y., Shiomi K., Hamada-Sato N. (2016). Reduction in IgE reactivity of pacific mackerel parvalbumin by heat treatment. Food Chemistry. 206: 78-84. [DOI: 10.1016/j.foodchem.2016.03.043] [DOI:10.1016/j.foodchem.2016.03.043] [PMID]
20. Kuniyoshi M.L.G., Da Silva-Gomes R.N., Zanella B.T.T., Dos Santos L.D., Dal-Pai-Silva M. (2019). Anabolism, catabolism and proteomic analysis in the slow-twitch muscle of pacu (Piaractus mesopotamicus) submitted to prolonged fasting followed by refeeding. Aquaculture Research. 51: 1101-1112. [DOI: 10.1111/are.14456] [DOI:10.1111/are.14456]
21. Laemmli U.K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 227: 680-685. [DOI:10.1038/227680a0] [PMID]
22. Lakmini K.P.C., Gonapinuwala S.T., Senarath H.P.S., Fernando C.A.N., Wijesekara I., De Croos M.D.S.T. (2022). Effect of autoclaving as a pre-treatment in the wet reduction process for extracting fish oil from yellowfin tuna heads. Sri Lanka Journal of Aquatic Sciences. 27: 43-61. [DOI: 10.4038/sljas.v27i1.7596] [DOI:10.4038/sljas.v27i1.7596]
23. Laly S.J., Sankar T.V., Panda S.K. (2022). Effect of pressure cooking alone and in combination with other treatments on shrimp allergic protein, tropomyosin. Journal of Food Science and Technology. 59: 1193-1201. [DOI: 10.1007/s13197-021-05124-2] [DOI:10.1007/s13197-021-05124-2] [PMID] [PMCID]
24. Li Q., Zhang L., Lu H., Song S., Luo Y. (2017). Comparison of postmortem changes in ATP-related compounds, protein degradation and endogenous enzyme activity of white muscle and dark muscle from common carp (Cyprinus carpio) stored at 4 °C. LWT. 78: 317-324. [DOI: 10.1016/j.lwt.2016.12.035] [DOI:10.1016/j.lwt.2016.12.035]
25. Li Z., Luo Y., Jiang M. (2016). Effects of heat treatment on the antigenicity and allergenicity of grass carp muscles. Journal of Aquatic Food Product Technology. 25: 350-357. [DOI: 10.1080/10498850.2013.851758] [DOI:10.1080/10498850.2013.851758]
26. Mardlijah S., Pane A.R.P., Fauzi M., Yusuf H.N., Widiyastuti H., Herlisman, Zamroni A., Noegroho T., Hufiadi, Wagiyo K. (2022). The fishing grounds and the exploitation status of Kawakawa (Euthynnus affinis) in Java sea, Indonesia. HAYATI Journal of Biosciences. 29: 255-265. [DOI: 10.4308/ hjb.29.2.255-265] [DOI:10.4308/hjb.29.2.255-265]
27. Mastrorilli C., Arasi S., Barni S., Caimmi D., Chiera F., Comberiati P., Dinardo G., Giannetti A., Gismondi M., Gracci S., Paravati F., Pelosi U., et al. (2023). IgE-mediated and non-IgE-mediated fish allergy in pediatric age: a holistic approach-a consensus by diagnostic commission of the Italian society of pediatric allergy and immunology. Medicina. 59: 1651. [DOI: 10.3390/ medicina59091651] [DOI:10.3390/medicina59091651] [PMID] [PMCID]
28. Maulidah R., Istyadji M., Sauqina. (2022). Pengaruh lama pemasakan yang berbeda terhadap kadar protein dan lemak tepung ikan papuyu (Anabas testudineus). JUSTER: Jurnal Sains dan Terapan. 1: 171-179. [DOI: 10.57218/juster.v1i3.370]. [Indonesian with English abstract] [DOI:10.57218/juster.v1i3.370]
29. Nguyen H.T., Hilmarsdóttir G.S., Tómasson T., Arason S., Gudjónsdóttir M. (2022). Changes in protein and non-protein nitrogen compounds during fishmeal processing-identification of unoptimized processing steps. Processes. 10: 621. [DOI: 10.3390/pr10040621] [DOI:10.3390/pr10040621]
30. Nugraha R., Ruethers T., Johnston E.B., Rolland J.M., O'hehir R.E., Kamath S.D., Lopata A.L. (2021). Effects of extraction buffer on the solubility and immunoreactivity of the pacific oyster allergens. Foods. 10: 16. [DOI: 10.3390/foods10020409] [DOI:10.3390/foods10020409] [PMID] [PMCID]
31. Ochiai Y., Ozawa H. (2020). Biochemical and physicochemical characteristics of the major muscle proteins from fish and shellfish. Fisheries Science. 86: 729-740. [DOI: 10.1007/s12562-020-01444-y] [DOI:10.1007/s12562-020-01444-y]
32. Palupi N.S., Indrastuti N.A., Wulandari N. (2021). Indonesian traditional salted fish: the alteration its allergenicity during processing. Journal of Aquatic Food Product Technology. 30: 353-363. [DOI: 10.1080/10498850.2021.1882632] [DOI:10.1080/10498850.2021.1882632]
33. Pramestia S.P., Riyanto B., Trilaksani W. (2015). Fish oil microencapsulation as omega-3 fatty acids fortification material for cream of crab soup. Jurnal Pengolahan Hasil Perikanan Indonesia. 18: 162-176. [DOI: 10.17844/jphpi.2015.18.2.162]. [Indonesian with English abstract] [DOI:10.17844/jphpi.2015.18.2.162]
34. Prester L. (2016). Seafood allergy, toxicity, and intolerance: a review. Journal of the American College of Nutrition. 35: 271-283. [DOI: 10.1080/07315724.2015.1014120] [DOI:10.1080/07315724.2015.1014120] [PMID]
35. Rittisak S., Charoen R., Savedboworn W. (2022). Broken riceberry (br) powder production using a double drum dryer and its utilization in the development of instant beverages. Processes. 10: 341. [DOI: 10.3390/pr10020341] [DOI:10.3390/pr10020341]
36. Ruethers T., Taki A.C., Johnston E.B., Nugraha R., Le T.T.K., Kalic T., McLean T.R., Kamath S.D., Lopata A.L. (2018). Seafood allergy: a comprehensive review of fish and shellfish allergens. Molecular Immunology. 100: 28-57. [DOI: 10.1016/j. molimm.2018.04.008] [DOI:10.1016/j.molimm.2018.04.008] [PMID]
37. Schrama D., Raposo De Magalhães C., Cerqueira M., Carrilho R., Revets D., Kuehn A., Engrola S., Rodrigues P.M. (2022). Fish processing and digestion affect parvalbumins detectability in gilthead seabream and European seabass. Animals. 12: 3022. [DOI: 10.3390/ani12213022] [DOI:10.3390/ani12213022] [PMID] [PMCID]
38. Soegiarto G., Abdullah M.S., Damayanti L.A., Suseno A., Effendi C. (2019). The prevalence of allergic diseases in school children of metropolitan city in Indonesia shows a similar pattern to that of developed countries. Asia Pacific Allergy. 9: e17. [DOI: 10.5415/apallergy.2019.9.e17] [DOI:10.5415/apallergy.2019.9.e17] [PMID] [PMCID]
39. Stevens J.R., Newton R.W., Tlusty M., Little D.C. (2018). The rise of aquaculture by-products: increasing food production, value, and sustainability through strategic utilisation. Marine Policy. 90: 115-124. [DOI: 10.1016/j.marpol.2017.12.027] [DOI:10.1016/j.marpol.2017.12.027]
40. Strasburg G., Xiong Y.L., Chiang W. (2017). Physiology and chemistry of edible muscle tissues. In: Damodaran S., Parkin K.L., Fennema O.R. (Editors). Fennema's food chemistry. 5th edition. CRC Press, Boca Raton. pp: 923-974. [DOI: 10.1201/9781315372914] [DOI:10.1201/9781315372914]
41. Tedner S.G., Asarnoj A., Thulin H., Westman M., Konradsen J.R., Nilsson C. (2022). Food allergy and hypersensitivity reactions in children and adults-a review. Journal of Internal Medicine. 291: 283-302. [DOI: 10.1111/joim.13422] [DOI:10.1111/joim.13422] [PMID]
42. Tiwari M., Barooah M.S., Sharma P., Bordoloi P.L., Hussain I.A., Ahmed A.M. (2021). Physico-chemical characteristics of fish flour prepared from locally available small indigenous fish species of Assam. Journal of Food Processing and Preservation. 45: e15704. [DOI: 10.1111/jfpp.15704] [DOI:10.1111/jfpp.15704]
43. Tontul I., Topuz A. (2017). Spray-drying of fruit and vegetable juices: Effect of drying conditions on the product yield and physical properties. Trends in Food Science and Technology. 63: 91-102. [DOI: 10.1016/j.tifs.2017.03.009] [DOI:10.1016/j.tifs.2017.03.009]
44. Tsai C.-L., Perng K., Hou Y.-C., Shen C.-J., Chen I.-N., Chen Y.-T. (2023). Effect of species, muscle location, food processing and refrigerated storage on the fish allergens, tropomyosin and parvalbumin. Food Chemistry. 402: 134479. [DOI: 10.1016/j.foodchem.2022.134479] [DOI:10.1016/j.foodchem.2022.134479] [PMID]
45. Vanga S.K., Singh A., Raghavan V. (2017). Review of conventional and novel food processing methods on food allergens. Critical Reviews in Food Science and Nutrition. 57: 2077-2094. [DOI: 10.1080/10408398.2015.1045965] [DOI:10.1080/10408398.2015.1045965] [PMID]
46. World Allergy Organization (WAO). (2013). WAO white book on allergy: update 2013, executive summary. WAO, USA. URL: https://allergypaais.org/wp-content/themes/twentytwentyone/ pdf/ExecSummary-2013-v6-hires.pdf.
47. Wulan N.L.N., Palupi N.S., Kusnandar F., Wulandari N. (2024). Autoclave process contribution to the production of black tiger shrimp powder (Penaeus monodon) and hypoallergenic puffing snacks. Jurnal Pengolahan Hasil Perikanan Indonesia. 27: 142-158. [DOI: 10.17844/jphpi.v27i2.50620]. [Indonesian with English abstract] [DOI:10.17844/jphpi.v27i2.50620]
48. Zhou F., He S., Sun H., Wang Y., Zhang Y. (2021). Advances in epitope mapping technologies for food protein allergens: a review. Trends in Food Science and Technology. 107: 226-239. [DOI: 10.1016/j.tifs.2020.10.035] [DOI:10.1016/j.tifs.2020.10.035]
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |







