Volume 12, Issue 2 (June 2025)
J. Food Qual. Hazards Control 2025, 12(2): 127-138 |
Back to browse issues page
Ethics code: Not applicable.
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Abrera G, Diano G, Tolentino M, Gragasin M, Cobar M, Caraos G, et al . Gamma Irradiation of Brown Rice: Effects of Dose, Variety, Paddy Age, and Packaging on Fungal Control, Shelf-Life and Post-Harvest Cost Estimate. J. Food Qual. Hazards Control 2025; 12 (2) :127-138
URL: http://jfqhc.ssu.ac.ir/article-1-1240-en.html
URL: http://jfqhc.ssu.ac.ir/article-1-1240-en.html
G.B. Abrera 

 , G. Diano
, G. Diano 
 , M.M. Tolentino
, M.M. Tolentino 
 , M.C.B. Gragasin
, M.C.B. Gragasin 
 , M.L.C. Cobar
, M.L.C. Cobar 
 , G. Caraos
, G. Caraos 
 , Z.M. De Guzman
, Z.M. De Guzman 
 , H.M. Solomon
, H.M. Solomon 
 , C.O. Asaad
, C.O. Asaad 
 , C.C. Deocaris *
, C.C. Deocaris * 




 , G. Diano
, G. Diano 
 , M.M. Tolentino
, M.M. Tolentino 
 , M.C.B. Gragasin
, M.C.B. Gragasin 
 , M.L.C. Cobar
, M.L.C. Cobar 
 , G. Caraos
, G. Caraos 
 , Z.M. De Guzman
, Z.M. De Guzman 
 , H.M. Solomon
, H.M. Solomon 
 , C.O. Asaad
, C.O. Asaad 
 , C.C. Deocaris *
, C.C. Deocaris * 


Biomedical Research Section, Department of Science and Technology – Philippine Nuclear Research Institute (DOST-PNRI), Commonwealth Avenue, Diliman, Quezon City, Philippines , ccdeocaris@pnri.dost.gov.ph
Full-Text [PDF 887 kb]
(543 Downloads)
| Abstract (HTML) (1287 Views)

Figure 3: Photographs of SL-218 grains in petri plates after inoculation of Aspergillus niger. Shown are results after zero, one, three, and six months of storage
Table 1: Effect of packaging material on Molds and Yeast Count (YMC) (log Colony Forming Units (CFU)/g) during long-term storage of RC-160 and SL-7 varieties*
Table 2: Effect of paddy age on Molds and Yeast Count (MYC) (log Colony Forming Units (CFU)/g) during long-term storage of brown rice *
Table 3: Impact of irradiation, storage, and maturity on brown rice moisture content*
Full-Text: (540 Views)
Gamma Irradiation of Brown Rice: Effects of Dose, Variety, Paddy Age, and Packaging on Fungal Control, Shelf-Life and Post-Harvest Cost Estimate
G.B. Abrera 1, G. Diano 1, M.M. Tolentino 1, M.C.B. Gragasin 3, M.L.C. Cobar 1, G. Caraos 1, Z.M. De Guzman 1, H.M. Solomon 2, C.O. Asaad 1, C.C. Deocaris 1[*]*
1. Biomedical Research Section, Department of Science and Technology – Philippine Nuclear Research Institute (DOST-PNRI), Commonwealth Avenue, Diliman, Quezon City, Philippines
2. Irradiation Services Section, Department of Science and Technology – Philippine Nuclear Research Institute (DOST-PNRI), Commonwealth Avenue, Diliman, Quezon City, Philippines
3. Philippine Center for Postharvest Development and Mechanization, Science City of Munoz, Nueva Ecija, Philippines
G.B. Abrera 1, G. Diano 1, M.M. Tolentino 1, M.C.B. Gragasin 3, M.L.C. Cobar 1, G. Caraos 1, Z.M. De Guzman 1, H.M. Solomon 2, C.O. Asaad 1, C.C. Deocaris 1[*]*
1. Biomedical Research Section, Department of Science and Technology – Philippine Nuclear Research Institute (DOST-PNRI), Commonwealth Avenue, Diliman, Quezon City, Philippines
2. Irradiation Services Section, Department of Science and Technology – Philippine Nuclear Research Institute (DOST-PNRI), Commonwealth Avenue, Diliman, Quezon City, Philippines
3. Philippine Center for Postharvest Development and Mechanization, Science City of Munoz, Nueva Ecija, Philippines
HIGHLIGHTS
- Gamma irradiation at 1 kGy significantly reduced mold and yeast in brown rice.
- The shelf-life of irradiated brown rice was extended up to eight months under ambient conditions.
- Neither the type of packaging material nor the age of the paddy significantly influenced the reduction of fungal load.
- Irradiation at 0.58 kGy achieves a 1-log reduction in Aspergillus niger contamination.
| Article type Original article |
ABSTRACT Background: Brown rice offers superior nutritional value compared to polished white rice but is less favored in rice-consuming countries due to its limited shelf-life. This study evaluates the efficacy of gamma irradiation in reducing fungal contamination in brown rice and extending its shelf-life, considering factors such as paddy age, rice variety, and packaging type. Methods: The efficacy of gamma irradiation (0-1 kGy) in reducing fungal contamination in brown rice was evaluated. RC-160 and SL-7 rice varieties (240 kg per variety) were harvested from Central Luzon (Region III) during the dry season (March–April 2023). One kg of two and eight-week-old paddy grains were packed in either Super Bag™, an International Rice Research Institute developed hermetic packaging, or conventional Polyethylene bags. Mold and Yeast Counts were quantified using 3M Petrifilm™ following Association of Official Agricultural Chemists official methods. Fifty g of each sample were homogenized in Butterfield’s phosphate buffer, serially diluted, plated, and incubated at 25±1 °C for 3-5 days. Statistical analyses were performed using one-way ANOVA and Tukey’s HSD post-hoc test with Statistical Tool for Agricultural Research (STAR) software, version 2.0.1 (IRRI). Results: Gamma irradiation at one kGy significantly (p<0.05) reduced Mold and Yeast Counts, with effects sustained for up to eight months. Paddy age and packaging type had no significant influence on fungal load. A dose of 0.58 kGy achieved a 1-log (90%) reduction in Aspergillus niger, while 3 kGy achieved complete inactivation, extending shelf-life up to six months. Cost analysis showed irradiation, logistics, and added storage fees increased retail price per kg by 0.14, 0.04, and US$0.02, respectively. Conclusions: Gamma irradiation effectively controls fungal contamination and extends brown rice shelf-life, potentially enhancing consumer confidence and promoting wider adoption of brown rice. © 2025, Shahid Sadoughi University of Medical Sciences. This is an open access article under the Creative Commons Attribution 4.0 International License. |
||
| Keywords Aspergillus niger Food Contamination Food Irradiation Food Packaging Radiation, Gamma |
|||
| Article history Received: 27 Nov 2024 Revised: 5 Feb 2025 Accepted: 30 Apr 2025 |
|||
| Abbreviations AOAC=Association of Official Agricultural Chemists CFU=Colony-Forming Unit IRRI=International Rice Research Institute MYC=Mold and Yeast Count PE=Polyethylene PhilMech=Philippine Center for Postharvest Development and Mechanization PNRI=Philippine Nuclear Research Institute SB=Super BagTM |
To cite: Abrera G.B., Diano G., Tolentino M.M., Gragasin M.C.B., Cobar M.L.C., Caraos G., De Guzman Z.M., Solomon H.M., Asaad C.O., Deocaris C.C. (2025). Gamma irradiation of brown rice: effects of dose, variety, paddy age, and packaging on fungal control, shelf-life and post-harvest cost estimate. Journal of Food Quality and Hazards Control. 12: 127-138.
Introduction
Introduction
Brown rice, a whole-grain alternative to polished white rice, has gained attention for its superior nutritional and functional properties, including higher levels of dietary fiber, essential fatty acids, vitamins, and bioactive compounds (Saleh et al., 2019). Despite these benefits, its consumption remains limited in rice-dependent countries like the Philippines, largely due to its poor storage stability, susceptibility to fungal contamination, and relatively short shelf-life (Borba et al., 2020). These limitations are particularly problematic in tropical climates where high humidity and temperature accelerate spoilage and microbial proliferation.
Fungal contamination is a major cause of postharvest loss in rice. Out of 24 identified rice grain diseases, 18 are fungal in origin, and postharvest losses in tropical regions can reach up to 50% (FAO, 2004; Kushiro, 2015). In addition to reducing grain quality and quantity, certain fungi, especially Aspergillus spp., produce mycotoxins such as aflatoxin B1, which pose serious food safety risks. These toxins are known for their hepatotoxic, carcinogenic, and immunosuppressive effects and have been found in rice at levels that sometimes exceed international safety limits (Eslami et al., 2015; Reddy et al., 2009). For example, local studies in the Philippines have reported a 95% incidence of aflatoxin B1 in brown and polished rice at a mean level of 1.53 µg/kg (Ali, 2019; Balendres et al., 2019; Sales and Yoshizawa, 2005).
Conventional approaches such as sun drying and hermetic storage have been used to minimize fungal growth, but these methods are often insufficient under challenging environmental conditions (Cinar and Onbaşı, 2020; Parfitt et al., 2010). As a result, food irradiation has been proposed as a supplementary postharvest intervention. Gamma irradiation offers a non-thermal, chemical-free technology for microbial decontamination and shelf-life extension (Mahajan et al., 2014; Sharma, 2004). Prior studies have demonstrated the effectiveness of gamma irradiation at doses of 1-10 kGy in reducing microbial loads in various food products, including rice, without significantly compromising sensory or nutritional quality (De Guzman et al., 1998; Lee and Kim, 2018; Nemtanu et al., 2005). However, most existing studies focus on polished rice or artificially inoculated samples, with limited attention to brown rice under practical storage conditions. Additionally, there is a lack of integrated research examining how factors such as rice variety, paddy age prior to milling, and packaging type influence the effectiveness of gamma irradiation in fungal control and shelf-life extension.
To address these gaps, this study investigates the effects of low-dose gamma irradiation (0-1 kGy) on the fungal load and shelf-life stability of brown rice. Specifically, it aims to (1) evaluate how radiation dose, rice variety, paddy age, and packaging material interact to influence fungal contamination, (2) determine the radiation dose required for Aspergillus niger inactivation, and (3) assess the cost implications of adopting irradiation as a postharvest treatment. Through this work, we aim to support the safe commercialization of brown rice by establishing a scientifically validated protocol for fungal control using gamma irradiation.
Materials and methods
Experimental design and phases
Experiments were performed in two phases to assess the effectiveness of ionizing radiation on enhancing shelf-life of brown rice by inhibiting fungal contamination. Phase I included an assessment of the microflora of brown rice and determination of radiation sensitivity of A. niger through an inoculation experiment. Phase II focused on a storage study of brown rice with a local warehouse and cost-benefit analysis.
Rice variety, sample collection, and packaging
To enhance the generalizability of findings across different genotypes, three rice varieties (RC-160, SL-7, and SL-218) were utilized in this study. SL-218 was used for fungal challenge and radio sensitivity testing to accurately determine the D10-value, while RC-160 and SL-7 were employed to assess storage stability under natural microbial conditions. Given that these were distinct experiments, the choice of rice genotype was dictated by variety availability. However, since the primary objective was to evaluate the radio sensitivity of A. niger in a controlled laboratory environment, the specific rice variety did not pose a major constraint. All the rice varieties were chosen due to their low amylose content, a trait commonly associated with desirable cooking and eating qualities, particularly a softer, more cohesive texture upon cooking (Glover et al., 2020; PhilRice, 2018). RC-160, also known as NSIC Rc 160, is a widely adopted cultivar developed by the Philippine Rice Research Institute (PhilRice), Science City of Muñoz, Nueva Ecija. It ranks among the top four preferred varieties in irrigated lowland systems, suitable for both dry and wet seasons. This variety exhibits an intermediate resistance profile against major rice pests and diseases, including rice blast, bacterial leaf blight, green leafhoppers, and stem borers (Glover et al., 2020). On the one end, SL-7 and SL-218 are classified as "super-hybrid" varieties developed by a private company. These hybrids are known for their high yield potential, superior grain quality, and broad-spectrum resistance to diseases and insect pests (Glover et al., 2020).
Procurement of the rice samples was carried out through institutional collaborations. RC-160 was obtained from the International Rice Research Institute (IRRI), while SL-7 and SL-218 were acquired through the Philippine Center for Postharvest Development and Mechanization (PhilMech) from a government-certified local rice trader based in Nueva Ecija.
For the storage stability component of the study, freshly harvested paddies (May and July 2015) were aged for either two or eight weeks prior to dehulling. Dehulling was performed using the PhilMech Compact Rice Huller (PhilMech, Philippines) to obtain brown rice. The dehulled grains were then packed at one kg per bag in two types of packaging materials: regular Polyethylene (PE) bags and hermetic Super Bags™ (SB). The SB, developed by IRRI and commercially produced by GrainPro, Inc. (Subic, Pampanga, Philippines), is specifically designed to minimize oxygen permeability and moisture ingress, thereby extending the shelf-life of stored grains (Kumar et al., 2020).
Preparation of A. niger suspension
A pure culture of A. niger ATCC 9029 was obtained from the culture collection of the Philippine National Collection of Microorganism (PNCM), BIOTECH, University of the Philippines-Los Baños, Laguna. A. niger was grown in Saboraud’s Dextrose Agar (SDA) medium (MH063, Hi-Media Lab Pvt. Ltd., USA) supplemented with 50 µg/L chloramphenicol (CO378-25G, Sigma Aldrich Co., USA) incubated at 30 oC for 5-7 days. The fungal inoculum was prepared by flooding the subculture plates with 0.05% Triton X-100 (X100-100ML, Sigma Aldrich Co., USA) and shaking with sterile glass beads to harvest spores. Spore concentration was estimated initially by adjusting the turbidity using sterile water and comparing the final inoculum with the McFarland standard to ensure a consistent and standardized bacterial inoculum density across all experiments. The actual concentration was later confirmed using a hemocytometer (Ingram and Bell Ltd., Germany). The suspension was adjusted as needed to achieve approximately 10⁸ Colony Forming Units (CFU)/ml, which served as the final inoculum for the succeeding experiments.
Fungal inoculation study
To evaluate the efficacy of gamma irradiation in eradicating fungal contamination in brown rice, challenge tests were conducted. Preceding A. niger inoculation, SL-218 grains underwent irradiation at 5 kGy, constituting a pre-sterilization dose. For each 25 g pack of pre-sterilized brown rice samples, two ml of freshly prepared A. niger was aseptically introduced, followed by manual mixing for 1-3 min to ensure uniform inoculation. Subsequently, the inoculated samples underwent a seven-day incubation period at 25 °C, followed by irradiation at varying absorbed doses (0.5, 1, 2, and 3 kGy). Enumeration of surviving A. niger was conducted using a rapid assay kit (PetrifilmTM, 3M Microbiology, USA) in accordance with Association of Official Agricultural Chemist official method 997.02 (AOAC, 2002). Results were expressed as log CFU/g of the sample.
Irradiation treatment
A total of 480 brown rice packs were prepared for the experiment, consisting of 240 packs stored each in PE and SB bags, with each pack containing one kg of brown rice. For irradiation, 10 individual packs were grouped together in ordinary plastic sacks, yielding 48 sacks in total. The sacks were distributed equally according to packaging type, radiation dose, and paddy age (two-week-old and eight-week-old samples). Each pair of sacks was placed in a metallic tote box (50×70×90 cm) for irradiation. Chemical dosimeters (B3 radiochromic film, GEX Corporation, USA) were placed at predetermined positions within the tote boxes to monitor and verify the minimum and maximum absorbed doses during the treatment process.
Irradiation was conducted using a Cobalt-60 source (activity=82.3 kCi) at the Multipurpose Irradiation Facility of the Philippine Nuclear Research Institute (PNRI), located in Diliman, Quezon City, Philippines. The samples were exposed to two radiation dose levels: 0.5 and 1 kGy. Dose selection was informed by prior studies, particularly that of Silos et al. (2024), which demonstrated a significant reduction in rancidity across RC-160, SL-7, and SL-218 brown rice varieties at 1 kGy. This dose was shown to effectively mitigate oxidative deterioration during storage, thereby complementing microbial decontamination efforts. Non-irradiated samples were included as controls to enable comparative analysis.
Immediately after irradiation, all treated and control samples were transported under ambient conditions to the PhilMech storage facility at the Science City of Muñoz, Nueva Ecija. The storage room was maintained at a controlled temperature of 26±1 °C and relative humidity of 70-75%. Environmental parameters were recorded daily throughout the storage period.
Microbiological assessment
To determine the fungal load, 50 g of each brown rice sample was aseptically transferred into a sterile Stomacher® blender bag with a filter (Interscience for Microbiology, France) containing 450 ml of sterile Butterfield’s phosphate buffer. The mixture was homogenized for 60 s using a paddle blender to ensure thorough suspension of surface microorganisms.
Serial decimal dilutions were subsequently prepared up to 1/105 by transferring 1 ml aliquots of the homogenate into sterile test tubes containing 9 ml of phosphate buffer, vortexed briefly to ensure uniform mixing. From each dilution, 1 ml was inoculated onto Mold and Yeast Count (MYC) Petrifilm™ rapid assay plates (3M Microbiology, USA), following the manufacturer’s protocol and the AOAC official method 997.02 (AOAC, 2002). The inoculated plates were incubated at 25 °C for three to five days.
Post-incubation, colonies were enumerated and expressed as CFU/g of sample. The fungal count for each treatment group was computed in accordance with the Bacteriological Analytical Manual (Tournas et al., 2001). All analyses were performed in triplicate. Data were reported in logarithmic (log₁₀ CFU/g) form to facilitate statistical interpretation. All the chemical reagents used in this study were analytical grade.
Post-harvest cost sensitivity analysis
A cost assessment was conducted to evaluate the economic implications of post-harvest treatments in brown rice, specifically focusing on storage duration, irradiation capacity, and postharvest losses. A One-at-A-Time (OAT) variation method was employed to systematically analyze the following cost components: (1) storage cost, (2) irradiation cost, (3) transportation cost, and (4) overall cost incorporating estimated postharvest losses with and without irradiation. Estimated unit cost based on exchange rate of US$1: PhP 56.27.
-Storage cost
Storage costs were calculated based on prevailing 50% warehouse utilization rates in Nueva Ecija, Philippines (Asia Rice Foundation, 2018; NFA, 2024). For control (non-irradiated) samples, a storage period of three months was adopted, aligning with spoilage thresholds established in previous work by Palomo et al. (2020). An extended storage period of eight months was also considered for irradiated samples, based on the observed stability and shelf-life improvements demonstrated in the current study.
-Irradiation cost
Irradiation cost was estimated with consideration of batch throughput, cobalt-60 source activity, and dose distribution parameters, based on the dose mapping outcomes from the present work. Cost modeling was conducted for two activity levels: 82.3 kCi (recorded in January 2015) and the current maximum of 155.3 kCi (as of January 2024), corresponding to the operational levels at the Multipurpose Irradiation Facility of the PNRI, Quezon City.
Irradiation logistics were modeled for a maximum of 42 tote boxes per day, with each tote box (70×50×90 cm) containing two sacks of ten one-kg brown rice packs. Each pack had approximate dimensions of 63×42×4.5 cm and a calculated density of 856.64 kg/m³. For conservative estimates, a lower throughput scenario of 36 tote boxes was also considered, consistent with previously validated operational thresholds. The Dose Uniformity Ratio (DUR) was assumed to fall within the range of 1.3-1.5, based on validated facility performance data for rice flour irradiation at comparable densities (~840 kg/m³). The institutionally approved irradiation cost at the Multipurpose Irradiation Facility is US$ 177.71 per run with a total of 14 tote boxes per run.
-Transportation cost
Transportation expenses were computed based on a round-trip transfer of brown rice batches between a designated local warehouse in the Science City of Muñoz, Nueva Ecija, and the PNRI irradiation facility in Quezon City, Metro Manila. Costs included vehicle fuel, labor, and handling fees for each batch cycle. The transportation cost was calculated using the prevailing local price of transporting rice to and from Nueva Ecija to Metro Manila, Philippines (US$ 0.89 per 50 kg, one-way trip).
-Postharvest losses
Postharvest losses were estimated by applying established fungal contamination rates, assuming spoilage due to Aspergillus spp. and other storage molds as the primary cause of quality degradation. The baseline fungal spoilage rate of 2.673% was adopted from the study by Castro (2003). Incremental costs associated with these losses were calculated following the economic impact model developed by Wongnaa et al. (2023), enabling a comparative assessment of total postharvest costs under irradiated versus non-irradiated storage conditions. The cost due to postharvest loss was calculated based on the calculated financial value of brown rice loss during storage following the prevailing local market price of brown rice at US$ 1.33 per kg.
Statistical analysis
To ensure normal distribution of the data for the statistical analysis, the raw data underwent transformation, either square root or tangent functions. Utilizing the IRRI Statistical Tool for Agricultural Research (STAR) version 2.0.1 (IRRI, 2014), significant differences among treatment groups across various factors were assessed through two-way ANOVA with Tukey's HSD Post Hoc Test (α=0.05). The radiation dose necessary for a 90% inactivation of A. niger (D10-value) was determined by plotting logarithms of the average log CFU against the corresponding radiation dose.
Results and discussion
Gamma irradiation significantly reduced fungal contamination in brown rice in a dose-dependent manner, supporting its potential as a postharvest intervention. The treatment effectively inactivated A. niger and maintained microbial quality within regulatory limits during storage. While rice variety, paddy age, and storage time influenced fungal load, packaging type had no significant effect. Cost analysis revealed that while irradiation and transport introduce additional expenses, these are offset by the reduced economic burden of postharvest losses. The following sections discuss these findings in greater detail, contextualizing them with existing literature and highlighting the implications for long-term storage and commercialization of irradiated brown rice.
Natural microflora of brown rice
The immediate post-treatment effect of gamma irradiation on the MYC of SL-218 brown rice was quantitatively assessed (Figure 1). The baseline MYC of untreated samples was 3.70±0.06 log CFU/g, which was significantly reduced to 1.00±0.01 log CFU/g following exposure to 3 kGy. A clear dose-dependent decline in fungal load was observed across the tested irradiation levels, with a pronounced reduction occurring at 1 kGy. At this dose, the MYC remained below the regulatory threshold established by the Philippine Food and Drug Administration (2022) for microbial safety in food products. Furthermore, samples treated with 2 and 3 kGy exhibited undetectable levels of fungal contamination, indicating complete suppression of viable molds and yeasts within the detection limits of the assay.

Figure 1: Comparison of Yeast and Mold Count (YMC) in gamma-irradiated brown rice across different radiation dose. Treatments sharing a letter showed no significant difference (p>0.05). CFU=Colony Forming Units
Radiation sensitivity of A. niger during storage
The inactivation kinetics of A. niger in brown rice were evaluated to determine the efficacy of gamma irradiation as a fungal control measure. A key parameter in microbial radio sensitivity studies, the D10-value, defined as the radiation dose required to achieve a one log (90%) reduction in viable microbial population, was experimentally determined to be 0.58 kGy. This value is indicative of moderate radio resistance and aligns with previous reports on filamentous fungal spores subjected to ionizing radiation (Cortésão et al., 2020; Koch et al., 2023).
Irradiation exhibited a dose-dependent inactivation effect on A. niger, with a high correlation between radiation dose and log reduction (R²=0.9116) (Figure 2). A dose of 1.74 kGy achieved a 3 log (99.9%) reduction, effectively rendering the fungal spores non-viable immediately post-treatment. This suggests that doses at or above this threshold may be adequate for initial decontamination of brown rice.
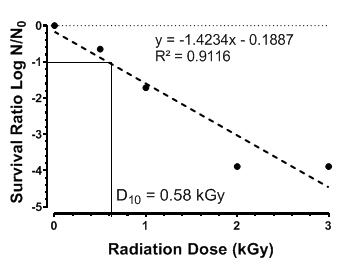
Figure 2: Survival curve of inoculated Aspergillus niger on SL-218 brown rice grains, illustrating the D10-value and the linear regression equation
Storage trials were conducted to evaluate the long-term antifungal efficacy of gamma irradiation in brown rice. As shown in Figure 3, non-irradiated control samples exhibited significant fungal proliferation after one month of storage under ambient conditions. In contrast, irradiated samples demonstrated dose-dependent fungal suppression over time. While initial fungal inhibition was observed across all irradiated groups, regrowth of A. niger became evident in samples treated with 0.5, 1, and 2 kGy after three and six months of storage, suggesting that these sublethal doses may not sustain long-term antifungal protection. Notably, samples irradiated at 3 kGy consistently exhibited undetectable levels of A. niger throughout the six-month storage period, highlighting the efficacy of this dose in achieving both immediate fungal inactivation and prolonged microbial stability.
Fungal contamination is a major cause of postharvest loss in rice. Out of 24 identified rice grain diseases, 18 are fungal in origin, and postharvest losses in tropical regions can reach up to 50% (FAO, 2004; Kushiro, 2015). In addition to reducing grain quality and quantity, certain fungi, especially Aspergillus spp., produce mycotoxins such as aflatoxin B1, which pose serious food safety risks. These toxins are known for their hepatotoxic, carcinogenic, and immunosuppressive effects and have been found in rice at levels that sometimes exceed international safety limits (Eslami et al., 2015; Reddy et al., 2009). For example, local studies in the Philippines have reported a 95% incidence of aflatoxin B1 in brown and polished rice at a mean level of 1.53 µg/kg (Ali, 2019; Balendres et al., 2019; Sales and Yoshizawa, 2005).
Conventional approaches such as sun drying and hermetic storage have been used to minimize fungal growth, but these methods are often insufficient under challenging environmental conditions (Cinar and Onbaşı, 2020; Parfitt et al., 2010). As a result, food irradiation has been proposed as a supplementary postharvest intervention. Gamma irradiation offers a non-thermal, chemical-free technology for microbial decontamination and shelf-life extension (Mahajan et al., 2014; Sharma, 2004). Prior studies have demonstrated the effectiveness of gamma irradiation at doses of 1-10 kGy in reducing microbial loads in various food products, including rice, without significantly compromising sensory or nutritional quality (De Guzman et al., 1998; Lee and Kim, 2018; Nemtanu et al., 2005). However, most existing studies focus on polished rice or artificially inoculated samples, with limited attention to brown rice under practical storage conditions. Additionally, there is a lack of integrated research examining how factors such as rice variety, paddy age prior to milling, and packaging type influence the effectiveness of gamma irradiation in fungal control and shelf-life extension.
To address these gaps, this study investigates the effects of low-dose gamma irradiation (0-1 kGy) on the fungal load and shelf-life stability of brown rice. Specifically, it aims to (1) evaluate how radiation dose, rice variety, paddy age, and packaging material interact to influence fungal contamination, (2) determine the radiation dose required for Aspergillus niger inactivation, and (3) assess the cost implications of adopting irradiation as a postharvest treatment. Through this work, we aim to support the safe commercialization of brown rice by establishing a scientifically validated protocol for fungal control using gamma irradiation.
Materials and methods
Experimental design and phases
Experiments were performed in two phases to assess the effectiveness of ionizing radiation on enhancing shelf-life of brown rice by inhibiting fungal contamination. Phase I included an assessment of the microflora of brown rice and determination of radiation sensitivity of A. niger through an inoculation experiment. Phase II focused on a storage study of brown rice with a local warehouse and cost-benefit analysis.
Rice variety, sample collection, and packaging
To enhance the generalizability of findings across different genotypes, three rice varieties (RC-160, SL-7, and SL-218) were utilized in this study. SL-218 was used for fungal challenge and radio sensitivity testing to accurately determine the D10-value, while RC-160 and SL-7 were employed to assess storage stability under natural microbial conditions. Given that these were distinct experiments, the choice of rice genotype was dictated by variety availability. However, since the primary objective was to evaluate the radio sensitivity of A. niger in a controlled laboratory environment, the specific rice variety did not pose a major constraint. All the rice varieties were chosen due to their low amylose content, a trait commonly associated with desirable cooking and eating qualities, particularly a softer, more cohesive texture upon cooking (Glover et al., 2020; PhilRice, 2018). RC-160, also known as NSIC Rc 160, is a widely adopted cultivar developed by the Philippine Rice Research Institute (PhilRice), Science City of Muñoz, Nueva Ecija. It ranks among the top four preferred varieties in irrigated lowland systems, suitable for both dry and wet seasons. This variety exhibits an intermediate resistance profile against major rice pests and diseases, including rice blast, bacterial leaf blight, green leafhoppers, and stem borers (Glover et al., 2020). On the one end, SL-7 and SL-218 are classified as "super-hybrid" varieties developed by a private company. These hybrids are known for their high yield potential, superior grain quality, and broad-spectrum resistance to diseases and insect pests (Glover et al., 2020).
Procurement of the rice samples was carried out through institutional collaborations. RC-160 was obtained from the International Rice Research Institute (IRRI), while SL-7 and SL-218 were acquired through the Philippine Center for Postharvest Development and Mechanization (PhilMech) from a government-certified local rice trader based in Nueva Ecija.
For the storage stability component of the study, freshly harvested paddies (May and July 2015) were aged for either two or eight weeks prior to dehulling. Dehulling was performed using the PhilMech Compact Rice Huller (PhilMech, Philippines) to obtain brown rice. The dehulled grains were then packed at one kg per bag in two types of packaging materials: regular Polyethylene (PE) bags and hermetic Super Bags™ (SB). The SB, developed by IRRI and commercially produced by GrainPro, Inc. (Subic, Pampanga, Philippines), is specifically designed to minimize oxygen permeability and moisture ingress, thereby extending the shelf-life of stored grains (Kumar et al., 2020).
Preparation of A. niger suspension
A pure culture of A. niger ATCC 9029 was obtained from the culture collection of the Philippine National Collection of Microorganism (PNCM), BIOTECH, University of the Philippines-Los Baños, Laguna. A. niger was grown in Saboraud’s Dextrose Agar (SDA) medium (MH063, Hi-Media Lab Pvt. Ltd., USA) supplemented with 50 µg/L chloramphenicol (CO378-25G, Sigma Aldrich Co., USA) incubated at 30 oC for 5-7 days. The fungal inoculum was prepared by flooding the subculture plates with 0.05% Triton X-100 (X100-100ML, Sigma Aldrich Co., USA) and shaking with sterile glass beads to harvest spores. Spore concentration was estimated initially by adjusting the turbidity using sterile water and comparing the final inoculum with the McFarland standard to ensure a consistent and standardized bacterial inoculum density across all experiments. The actual concentration was later confirmed using a hemocytometer (Ingram and Bell Ltd., Germany). The suspension was adjusted as needed to achieve approximately 10⁸ Colony Forming Units (CFU)/ml, which served as the final inoculum for the succeeding experiments.
Fungal inoculation study
To evaluate the efficacy of gamma irradiation in eradicating fungal contamination in brown rice, challenge tests were conducted. Preceding A. niger inoculation, SL-218 grains underwent irradiation at 5 kGy, constituting a pre-sterilization dose. For each 25 g pack of pre-sterilized brown rice samples, two ml of freshly prepared A. niger was aseptically introduced, followed by manual mixing for 1-3 min to ensure uniform inoculation. Subsequently, the inoculated samples underwent a seven-day incubation period at 25 °C, followed by irradiation at varying absorbed doses (0.5, 1, 2, and 3 kGy). Enumeration of surviving A. niger was conducted using a rapid assay kit (PetrifilmTM, 3M Microbiology, USA) in accordance with Association of Official Agricultural Chemist official method 997.02 (AOAC, 2002). Results were expressed as log CFU/g of the sample.
Irradiation treatment
A total of 480 brown rice packs were prepared for the experiment, consisting of 240 packs stored each in PE and SB bags, with each pack containing one kg of brown rice. For irradiation, 10 individual packs were grouped together in ordinary plastic sacks, yielding 48 sacks in total. The sacks were distributed equally according to packaging type, radiation dose, and paddy age (two-week-old and eight-week-old samples). Each pair of sacks was placed in a metallic tote box (50×70×90 cm) for irradiation. Chemical dosimeters (B3 radiochromic film, GEX Corporation, USA) were placed at predetermined positions within the tote boxes to monitor and verify the minimum and maximum absorbed doses during the treatment process.
Irradiation was conducted using a Cobalt-60 source (activity=82.3 kCi) at the Multipurpose Irradiation Facility of the Philippine Nuclear Research Institute (PNRI), located in Diliman, Quezon City, Philippines. The samples were exposed to two radiation dose levels: 0.5 and 1 kGy. Dose selection was informed by prior studies, particularly that of Silos et al. (2024), which demonstrated a significant reduction in rancidity across RC-160, SL-7, and SL-218 brown rice varieties at 1 kGy. This dose was shown to effectively mitigate oxidative deterioration during storage, thereby complementing microbial decontamination efforts. Non-irradiated samples were included as controls to enable comparative analysis.
Immediately after irradiation, all treated and control samples were transported under ambient conditions to the PhilMech storage facility at the Science City of Muñoz, Nueva Ecija. The storage room was maintained at a controlled temperature of 26±1 °C and relative humidity of 70-75%. Environmental parameters were recorded daily throughout the storage period.
Microbiological assessment
To determine the fungal load, 50 g of each brown rice sample was aseptically transferred into a sterile Stomacher® blender bag with a filter (Interscience for Microbiology, France) containing 450 ml of sterile Butterfield’s phosphate buffer. The mixture was homogenized for 60 s using a paddle blender to ensure thorough suspension of surface microorganisms.
Serial decimal dilutions were subsequently prepared up to 1/105 by transferring 1 ml aliquots of the homogenate into sterile test tubes containing 9 ml of phosphate buffer, vortexed briefly to ensure uniform mixing. From each dilution, 1 ml was inoculated onto Mold and Yeast Count (MYC) Petrifilm™ rapid assay plates (3M Microbiology, USA), following the manufacturer’s protocol and the AOAC official method 997.02 (AOAC, 2002). The inoculated plates were incubated at 25 °C for three to five days.
Post-incubation, colonies were enumerated and expressed as CFU/g of sample. The fungal count for each treatment group was computed in accordance with the Bacteriological Analytical Manual (Tournas et al., 2001). All analyses were performed in triplicate. Data were reported in logarithmic (log₁₀ CFU/g) form to facilitate statistical interpretation. All the chemical reagents used in this study were analytical grade.
Post-harvest cost sensitivity analysis
A cost assessment was conducted to evaluate the economic implications of post-harvest treatments in brown rice, specifically focusing on storage duration, irradiation capacity, and postharvest losses. A One-at-A-Time (OAT) variation method was employed to systematically analyze the following cost components: (1) storage cost, (2) irradiation cost, (3) transportation cost, and (4) overall cost incorporating estimated postharvest losses with and without irradiation. Estimated unit cost based on exchange rate of US$1: PhP 56.27.
-Storage cost
Storage costs were calculated based on prevailing 50% warehouse utilization rates in Nueva Ecija, Philippines (Asia Rice Foundation, 2018; NFA, 2024). For control (non-irradiated) samples, a storage period of three months was adopted, aligning with spoilage thresholds established in previous work by Palomo et al. (2020). An extended storage period of eight months was also considered for irradiated samples, based on the observed stability and shelf-life improvements demonstrated in the current study.
-Irradiation cost
Irradiation cost was estimated with consideration of batch throughput, cobalt-60 source activity, and dose distribution parameters, based on the dose mapping outcomes from the present work. Cost modeling was conducted for two activity levels: 82.3 kCi (recorded in January 2015) and the current maximum of 155.3 kCi (as of January 2024), corresponding to the operational levels at the Multipurpose Irradiation Facility of the PNRI, Quezon City.
Irradiation logistics were modeled for a maximum of 42 tote boxes per day, with each tote box (70×50×90 cm) containing two sacks of ten one-kg brown rice packs. Each pack had approximate dimensions of 63×42×4.5 cm and a calculated density of 856.64 kg/m³. For conservative estimates, a lower throughput scenario of 36 tote boxes was also considered, consistent with previously validated operational thresholds. The Dose Uniformity Ratio (DUR) was assumed to fall within the range of 1.3-1.5, based on validated facility performance data for rice flour irradiation at comparable densities (~840 kg/m³). The institutionally approved irradiation cost at the Multipurpose Irradiation Facility is US$ 177.71 per run with a total of 14 tote boxes per run.
-Transportation cost
Transportation expenses were computed based on a round-trip transfer of brown rice batches between a designated local warehouse in the Science City of Muñoz, Nueva Ecija, and the PNRI irradiation facility in Quezon City, Metro Manila. Costs included vehicle fuel, labor, and handling fees for each batch cycle. The transportation cost was calculated using the prevailing local price of transporting rice to and from Nueva Ecija to Metro Manila, Philippines (US$ 0.89 per 50 kg, one-way trip).
-Postharvest losses
Postharvest losses were estimated by applying established fungal contamination rates, assuming spoilage due to Aspergillus spp. and other storage molds as the primary cause of quality degradation. The baseline fungal spoilage rate of 2.673% was adopted from the study by Castro (2003). Incremental costs associated with these losses were calculated following the economic impact model developed by Wongnaa et al. (2023), enabling a comparative assessment of total postharvest costs under irradiated versus non-irradiated storage conditions. The cost due to postharvest loss was calculated based on the calculated financial value of brown rice loss during storage following the prevailing local market price of brown rice at US$ 1.33 per kg.
Statistical analysis
To ensure normal distribution of the data for the statistical analysis, the raw data underwent transformation, either square root or tangent functions. Utilizing the IRRI Statistical Tool for Agricultural Research (STAR) version 2.0.1 (IRRI, 2014), significant differences among treatment groups across various factors were assessed through two-way ANOVA with Tukey's HSD Post Hoc Test (α=0.05). The radiation dose necessary for a 90% inactivation of A. niger (D10-value) was determined by plotting logarithms of the average log CFU against the corresponding radiation dose.
Results and discussion
Gamma irradiation significantly reduced fungal contamination in brown rice in a dose-dependent manner, supporting its potential as a postharvest intervention. The treatment effectively inactivated A. niger and maintained microbial quality within regulatory limits during storage. While rice variety, paddy age, and storage time influenced fungal load, packaging type had no significant effect. Cost analysis revealed that while irradiation and transport introduce additional expenses, these are offset by the reduced economic burden of postharvest losses. The following sections discuss these findings in greater detail, contextualizing them with existing literature and highlighting the implications for long-term storage and commercialization of irradiated brown rice.
Natural microflora of brown rice
The immediate post-treatment effect of gamma irradiation on the MYC of SL-218 brown rice was quantitatively assessed (Figure 1). The baseline MYC of untreated samples was 3.70±0.06 log CFU/g, which was significantly reduced to 1.00±0.01 log CFU/g following exposure to 3 kGy. A clear dose-dependent decline in fungal load was observed across the tested irradiation levels, with a pronounced reduction occurring at 1 kGy. At this dose, the MYC remained below the regulatory threshold established by the Philippine Food and Drug Administration (2022) for microbial safety in food products. Furthermore, samples treated with 2 and 3 kGy exhibited undetectable levels of fungal contamination, indicating complete suppression of viable molds and yeasts within the detection limits of the assay.

Figure 1: Comparison of Yeast and Mold Count (YMC) in gamma-irradiated brown rice across different radiation dose. Treatments sharing a letter showed no significant difference (p>0.05). CFU=Colony Forming Units
Radiation sensitivity of A. niger during storage
The inactivation kinetics of A. niger in brown rice were evaluated to determine the efficacy of gamma irradiation as a fungal control measure. A key parameter in microbial radio sensitivity studies, the D10-value, defined as the radiation dose required to achieve a one log (90%) reduction in viable microbial population, was experimentally determined to be 0.58 kGy. This value is indicative of moderate radio resistance and aligns with previous reports on filamentous fungal spores subjected to ionizing radiation (Cortésão et al., 2020; Koch et al., 2023).
Irradiation exhibited a dose-dependent inactivation effect on A. niger, with a high correlation between radiation dose and log reduction (R²=0.9116) (Figure 2). A dose of 1.74 kGy achieved a 3 log (99.9%) reduction, effectively rendering the fungal spores non-viable immediately post-treatment. This suggests that doses at or above this threshold may be adequate for initial decontamination of brown rice.

Figure 2: Survival curve of inoculated Aspergillus niger on SL-218 brown rice grains, illustrating the D10-value and the linear regression equation
Storage trials were conducted to evaluate the long-term antifungal efficacy of gamma irradiation in brown rice. As shown in Figure 3, non-irradiated control samples exhibited significant fungal proliferation after one month of storage under ambient conditions. In contrast, irradiated samples demonstrated dose-dependent fungal suppression over time. While initial fungal inhibition was observed across all irradiated groups, regrowth of A. niger became evident in samples treated with 0.5, 1, and 2 kGy after three and six months of storage, suggesting that these sublethal doses may not sustain long-term antifungal protection. Notably, samples irradiated at 3 kGy consistently exhibited undetectable levels of A. niger throughout the six-month storage period, highlighting the efficacy of this dose in achieving both immediate fungal inactivation and prolonged microbial stability.

Figure 3: Photographs of SL-218 grains in petri plates after inoculation of Aspergillus niger. Shown are results after zero, one, three, and six months of storage
Interestingly, the fungal load in non-irradiated control samples at six months appeared lower than that at one and three months, while the fungal growth in 2 kGy-treated samples exceeded that of the control at the same time point. This counterintuitive trend may be partially explained by changes in spore viability and colonization behavior over time. It aligns with recent findings that spore density significantly influences A. niger germination, wherein excessively high spore loads can inhibit germination due to quorum-sensing-like mechanisms or competitive interactions (Ijadpanahsaravi et al., 2024). Given that ionizing radiation may alter spore viability and density distribution during storage, such interactions could affect the dynamics of fungal resurgence at sublethal doses. These results collectively support the conclusion that a minimum dose of 3 kGy is necessary to ensure effective and sustained suppression of fungal contaminants in brown rice under ambient storage conditions.
The observed variability in fungal resistance during storage may be attributed to protective structural and biochemical features of A. niger spores. Fungal spores possess multilayered cell walls composed of polysaccharides, such as chitin and glucans, and are enveloped by an outer hydrophobic layer rich in rodlet-forming proteins (hydrophobins) and protective pigments like melanin. These structures confer resilience by shielding intracellular components, particularly DNA, from radiation-induced damage and oxidative stress mediated by reactive oxygen species (Cortésão et al., 2020; Koch et al., 2023). Such features may partially account for the limited efficacy of lower radiation doses over extended storage durations.
Rice variety, packaging, and paddy age effects
Fungal colonization in grains is influenced not only by spore survival but also by successful adherence, germination, and proliferation under conducive environmental conditions. These include temperature, water activity, light availability, nutrient content, and, notably, spore density (Ijadpanahsaravi et al., 2024). Importantly, the substrate characteristics and their interaction with environmental variables must be evaluated to simulate real-world storage conditions. Recognizing that grain quality is affected by environmental factors during cultivation, postharvest handling, and processing, we examined the role of paddy age defined as the time elapsed between harvest and dehulling on fungal susceptibility in brown rice (IRRI, 2024).
Effect of rice variety. Two brown rice varieties (RC-160 and SL-7) were assessed to evaluate varietal differences in susceptibility to fungal contamination. Statistical analysis revealed a highly significant difference in mean MYC between the two varieties (p<0.001), with SL-7 consistently exhibiting lower fungal loads compared to RC-160 (Table 1). This variation may be attributed to intrinsic varietal characteristics, as well as differences in environmental conditions during cultivation, post-harvest handling, and storage practices. At an irradiation dose of one kGy, MYC levels in both varieties remained within acceptable regulatory limits (≤2 log CFU/g) for up to eight months under ambient storage conditions, as defined by the Philippine Food and Drug Administration (2022). Moreover, increasing the irradiation dose beyond one kGy resulted in a statistically significant reduction in fungal load in both varieties (p<0.001), indicating a dose-dependent antifungal effect of gamma irradiation.
The observed variability in fungal resistance during storage may be attributed to protective structural and biochemical features of A. niger spores. Fungal spores possess multilayered cell walls composed of polysaccharides, such as chitin and glucans, and are enveloped by an outer hydrophobic layer rich in rodlet-forming proteins (hydrophobins) and protective pigments like melanin. These structures confer resilience by shielding intracellular components, particularly DNA, from radiation-induced damage and oxidative stress mediated by reactive oxygen species (Cortésão et al., 2020; Koch et al., 2023). Such features may partially account for the limited efficacy of lower radiation doses over extended storage durations.
Rice variety, packaging, and paddy age effects
Fungal colonization in grains is influenced not only by spore survival but also by successful adherence, germination, and proliferation under conducive environmental conditions. These include temperature, water activity, light availability, nutrient content, and, notably, spore density (Ijadpanahsaravi et al., 2024). Importantly, the substrate characteristics and their interaction with environmental variables must be evaluated to simulate real-world storage conditions. Recognizing that grain quality is affected by environmental factors during cultivation, postharvest handling, and processing, we examined the role of paddy age defined as the time elapsed between harvest and dehulling on fungal susceptibility in brown rice (IRRI, 2024).
Effect of rice variety. Two brown rice varieties (RC-160 and SL-7) were assessed to evaluate varietal differences in susceptibility to fungal contamination. Statistical analysis revealed a highly significant difference in mean MYC between the two varieties (p<0.001), with SL-7 consistently exhibiting lower fungal loads compared to RC-160 (Table 1). This variation may be attributed to intrinsic varietal characteristics, as well as differences in environmental conditions during cultivation, post-harvest handling, and storage practices. At an irradiation dose of one kGy, MYC levels in both varieties remained within acceptable regulatory limits (≤2 log CFU/g) for up to eight months under ambient storage conditions, as defined by the Philippine Food and Drug Administration (2022). Moreover, increasing the irradiation dose beyond one kGy resulted in a statistically significant reduction in fungal load in both varieties (p<0.001), indicating a dose-dependent antifungal effect of gamma irradiation.
Table 1: Effect of packaging material on Molds and Yeast Count (YMC) (log Colony Forming Units (CFU)/g) during long-term storage of RC-160 and SL-7 varieties*
| Storage Time (months) | |||||||||||||||||||
| 0 | 2 | 4 | 6 | 8 | |||||||||||||||
| RC-160 | Packaging Material |
PE | SB | PE | SB | PE | SB | PE | SB | PE | SB | ||||||||
| Dose (kGy) | |||||||||||||||||||
| Control | 3.23±0.11 Aa | 3.22±0.13 Aa | 3.26±0.27 Aa | 3.22±0.22 Aa | 2.90± 0.06 Aa | 2.87±0.08 Aa | 2.65±0.15 Aa | 2.59±0.14 Aa | 2.29±0.10 Aa | 2.23±0.16 Aa | |||||||||
| 0.5 | 3.09±0.07 Ab | 3.10±0.07 Ab | 3.02±0.15Ab | 2.97±0.08 Ab | 2.79±0.08 Ab | 2.76±0.11 Ab | 2.49±0.21 Aa | 2.48±0.22 Aa | 2.11±0.07 Bb | 2.29±0.13 Aa | |||||||||
| 1.0 | 2.91±0.03 Ac | 2.93±0.02 Ac | 2.91±0.11 Ab | 2.92±0.10 Ab | 2.69±0.10 Ac | 2.67±0.12 Ac | 2.43±0.26 Aa | 2.45±0.24 Aa | 2.23±0.19 Aa | 2.00±0.00 Bb | |||||||||
| SL-7 | Control | 2.78±0.09 Aa | 2.67±0.09 Ba | 2.98±0.26 Aa | 2.89±0.17 Aa | 2.80±0.04 Aa | 2.76±0.05 Aa | 2.61±0.10 Aa | 2.67±0.09 Aa | 2.43±0.09 Aa | 2.24±0.25 Aa | ||||||||
| 0.5 | 2.71±0.10 Aa | 2.63±0.11 Ba | 2.69±0.11 Ab | 2.72±0.04 Ab | 2.76±0.07 Aa | 2.72±0.03 Aa | 2.63±0.11 Aa | 2.60±0.05 Aa | 2.27±0.13 Aa | 2.22±0.25 Aa | |||||||||
| 1.0 | 2.67±0.14 Aa | 2.55±0.14 Ba | 2.68±0.09 Ab | 2.77±0.08 Ab | 2.67±0.07 Ab | 2.63±0.11 Ab | 2.57±0.06 Aa | 2.61±0.08 Aa | 2.19±0.21 Aa | 2.25±0.27 Aa | |||||||||
* Data shows the long-term storage effects of packaging material (IRRI Super Bag™ (SB) vs. standard Polyethylene (PE) bags on MYC in two rice varieties (RC-160 and SL-7). A two-way ANOVA with Tukey's HSD post hoc analysis was performed for each storage time and variety. Data are presented as Mean±SD (n=6). Different superscripts (column-wise uppercase, row-wise lowercase) indicate significant differences (p<0.05).
Effect of packaging type. Previous studies have shown that the type of packaging material can significantly influence the physicochemical stability and microbiological quality of stored grains (Ramezanzadeh et al., 2000). These effects are often attributed to chemical reactions associated with lipid oxidation and rancidity, which are influenced by the internal microenvironment, particularly oxygen and moisture levels, within the packaging. In this context, a comparative microbiological assessment was conducted on brown rice packaged in conventional PE and hermetic SB bags. The influence of packaging type on MYC was evaluated in both irradiated and non-irradiated samples of SL-218 and RC-160 varieties across multiple storage intervals (Table 1).
While gamma irradiation led to a statistically significant reduction in MYC in a dose-dependent manner (p<0.0001), no significant difference was observed between PE bags and SB (p=0.445). At an irradiation dose of one kGy, MYC levels were reduced to within acceptable regulatory limits immediately post-treatment and remained consistently low throughout the storage period, irrespective of packaging material. This finding suggests that the antifungal efficacy of gamma irradiation at this dose may override any supplementary protective benefits offered by SB packaging in terms of fungal control.
These results are consistent with prior findings by Boonchoo et al. (2005) and Shobha et al. (2024), which demonstrated that irradiation at one kGy was sufficient to inhibit fungal proliferation across various grain types, regardless of packaging type. Although SB packaging is known to limit gas exchange and maintain grain moisture content more effectively than conventional materials (Sorn et al., 2017), its impact on MYC reduction appears minimal under the conditions tested. It was also noted by Sorn et al. (2017) that there is an increased susceptibility to fungal contamination in rice stored in SB with an initial moisture content of 16%, compared to samples with 14%. In the present study, the moisture content of both brown rice varieties remained below 14%, suggesting that differences in MYC cannot be attributed to superior hermeticity of SB over PE bags.
Effect of paddy age. The influence of paddy age on MYC was evaluated over an eight-month storage period (Table 2). Statistically significant differences were observed between two- and 8-week-old paddies, with the former consistently exhibiting higher MYC (p=0.03). This trend persisted across all radiation doses and storage durations, becoming more pronounced at higher doses and longer storage periods. Notably, immediately after irradiation, the two-week-old RC-160 paddy had higher MYC than its eight-week counterpart (p<0.0001), despite both paddy samples exhibiting moisture contents below the 14% safe storage threshold (p=0.0472) (BAFPS, 2014).
While gamma irradiation led to a statistically significant reduction in MYC in a dose-dependent manner (p<0.0001), no significant difference was observed between PE bags and SB (p=0.445). At an irradiation dose of one kGy, MYC levels were reduced to within acceptable regulatory limits immediately post-treatment and remained consistently low throughout the storage period, irrespective of packaging material. This finding suggests that the antifungal efficacy of gamma irradiation at this dose may override any supplementary protective benefits offered by SB packaging in terms of fungal control.
These results are consistent with prior findings by Boonchoo et al. (2005) and Shobha et al. (2024), which demonstrated that irradiation at one kGy was sufficient to inhibit fungal proliferation across various grain types, regardless of packaging type. Although SB packaging is known to limit gas exchange and maintain grain moisture content more effectively than conventional materials (Sorn et al., 2017), its impact on MYC reduction appears minimal under the conditions tested. It was also noted by Sorn et al. (2017) that there is an increased susceptibility to fungal contamination in rice stored in SB with an initial moisture content of 16%, compared to samples with 14%. In the present study, the moisture content of both brown rice varieties remained below 14%, suggesting that differences in MYC cannot be attributed to superior hermeticity of SB over PE bags.
Effect of paddy age. The influence of paddy age on MYC was evaluated over an eight-month storage period (Table 2). Statistically significant differences were observed between two- and 8-week-old paddies, with the former consistently exhibiting higher MYC (p=0.03). This trend persisted across all radiation doses and storage durations, becoming more pronounced at higher doses and longer storage periods. Notably, immediately after irradiation, the two-week-old RC-160 paddy had higher MYC than its eight-week counterpart (p<0.0001), despite both paddy samples exhibiting moisture contents below the 14% safe storage threshold (p=0.0472) (BAFPS, 2014).
Table 2: Effect of paddy age on Molds and Yeast Count (MYC) (log Colony Forming Units (CFU)/g) during long-term storage of brown rice *
| Storage Months | |||||||||||
| 0 | 2 | 4 | 6 | 8 | |||||||
| Paddy Age | 2W | 8W | 2W | 8W | 2W | 8W | 2W | 8W | 2W | 8W | |
| RC-160 | Dose (kGy) | ||||||||||
| Control | 3.32±0.07 Aa | 3.13±0.07 Ba | 3.46±0.07 Aa | 3.01±0.04 Ba | 2.94±0.03 Aa | 2.82±0.04 Ba | 2.75±0.06 Aa | 2.50±0.05 Ba | 2.21±0.14 Aa | 2.32±0.12 Aa | |
| 0.5 | 3.14±0.05 Ab | 3.05±0.04 Bb | 3.09±0.07 Ab | 2.90±0.05 Bb | 2.84±0.08 Ab | 2.71±0.06 Bb | 2.67±0.06 Ab | 2.30±0.04 Bb | 2.26±0.16 Ab | 2.15±0.09 Ab | |
| 1.0 | 2.90±0.03 Ac | 2.93±0.01 Ac | 3.01±0.02 Ac | 2.82±0.04 Bc | 2.77±0.05 Ac | 2.60±0.07 Bc | 2.66±0.04 Ab | 2.22±0.08 Bc | 2.20±0.22 Ac | 2.03±0.05 Ac | |
| SL-7 | Control | 2.73±0.03 Aa | 2.73±0.15 Aa | 3.13±0.12 Aa | 2.75±0.01 Ba | 2.79±0.04 Aa | 2.77±0.06 Aa | 2.72±0.03 Aa | 2.56±0.06 Ba | 2.20±0.21 Ba | 2.47±0.06 Aa |
| 0.5 | 2.66±0.12 Aa | 2.68±0.11 Aa | 2.67±0.10 Ab | 2.75±0.03 Aa | 2.76±0.06 Ab | 2.72±0.03 Aa | 2.69±0.05 Ab | 2.54±0.03 Bb | 2.08±0.10 Bb | 2.41±0.08 Ab | |
| 1.0 | 2.49±0.08 Bb | 2.73±0.09 Aa | 2.67±0.08 Bb | 2.78±0.07 Aa | 2.73±0.05 Ab | 2.58±0.05 Bb | 2.64±0.04 Ab | 2.54±0.04 Bb | 2.00±0.00 Bb | 2.44±0.07 Ab | |
* Data shows the long-term storage effects of paddy age on MYC in two rice varieties: RC-160 and SL-7. A two-way ANOVA with Tukey's HSD post-hoc analysis was performed. Data are presented as Mean±SD (n=6 observations per dose at each storage period). Means with different superscripts (uppercase for column-wise, lowercase for row-wise) are significantly different (p<0.05) at a 95% confidence level.
Interestingly, higher MYC in the two-week-old paddy was observed despite expectations that the eight-week-old paddy, with slightly higher moisture, would exhibit increased fungal susceptibility. Moreover, the divergence in MYC between paddy ages was negligible in non-irradiated and 0.5 kGy-irradiated samples, suggesting that the initial microbial load plays a more significant role at lower radiation doses. Irradiation at higher doses (≥1 kGy) effectively mitigated these age-related differences by reducing viable spore counts.
Storage of rice paddies is a customary practice among countries where the crop is a staple food and where seasonal harvest may limit its supply during the off-season (Saikrishna et al., 2018; Zhou et al., 2002). However, during storage, the natural process of rice aging ensues, mainly due to changes that occur in its chemical composition, such as starch, protein, and lipid contents, which then influence its physical properties, like water absorption, volume expansion in cooking, hardness, and adhesiveness (Zhou et al., 2015). Most notable among these was the increased water absorption required to cook aged rice, which peaks at two to four months after harvest and then declines to baseline values afterward (Chrastil, 1990; Faruq et al., 2015; Peng et al., 2019; Swamy et al., 1978). This behavior is attributed to the strengthening of the crystalline structure of amylose during storage resulting from the action of debranching enzymes that make it densely packed and harder for water to infiltrate. Contributing to an increase in water impenetrability are the oxidation of proteins into disulfide bonds and lipids into free fatty acids during storage, which can form a strong network around starch (Peng et al., 2019; Saikrishna et al., 2018). Considering that fungi thrive on warmth, moisture, and an acidic environment and that the storage temperature was controlled for both types of paddies, it may be inferred that moisture or pH differences could have promoted fungal growth on two-week-old paddies. Eight-week-old paddies, being more water-impermeable due to the structural and chemical changes of aging, may be a less suitable substrate for molds compared with the two-week-old paddy.
Cost-benefit analysis of gamma irradiation for brown rice storage stability
Table 4 summarizes the estimated incremental costs (in USD) associated with irradiation, storage, and transportation of brown rice samples. The additional expenditures incurred per kilogram were US$ 0.02 for storage, at least US$ 0.14 for irradiation, and US$ 0.04 for transportation, relative to the prevailing market price of brown rice in the Philippines. These figures correspond to a 1.50, 10.53, and 3.01% increase, respectively. In comparison, the estimated cost of mitigating postharvest losses through conventional means amounted to US$ 0.40 per kilogram, reflecting a substantial 30.08% increase in total cost.
From an economic standpoint, while irradiation contributes to marginal increases in storage and transportation costs primarily due to extended shelf-life and logistical handling the overall financial impact is offset by the marked reduction in postharvest losses. Interestingly, based on the economic model used in this study, although initial investments in irradiation infrastructure and operations may appear significant, irradiation resulted in a 30% reduction in economic losses associated with spoilage and fungal contamination during storage of brown rice. Thus, the long-term cost savings due to reduced product loss substantiate the economic viability of food irradiation as a postharvest intervention strategy if scaled at a national level.
Storage of rice paddies is a customary practice among countries where the crop is a staple food and where seasonal harvest may limit its supply during the off-season (Saikrishna et al., 2018; Zhou et al., 2002). However, during storage, the natural process of rice aging ensues, mainly due to changes that occur in its chemical composition, such as starch, protein, and lipid contents, which then influence its physical properties, like water absorption, volume expansion in cooking, hardness, and adhesiveness (Zhou et al., 2015). Most notable among these was the increased water absorption required to cook aged rice, which peaks at two to four months after harvest and then declines to baseline values afterward (Chrastil, 1990; Faruq et al., 2015; Peng et al., 2019; Swamy et al., 1978). This behavior is attributed to the strengthening of the crystalline structure of amylose during storage resulting from the action of debranching enzymes that make it densely packed and harder for water to infiltrate. Contributing to an increase in water impenetrability are the oxidation of proteins into disulfide bonds and lipids into free fatty acids during storage, which can form a strong network around starch (Peng et al., 2019; Saikrishna et al., 2018). Considering that fungi thrive on warmth, moisture, and an acidic environment and that the storage temperature was controlled for both types of paddies, it may be inferred that moisture or pH differences could have promoted fungal growth on two-week-old paddies. Eight-week-old paddies, being more water-impermeable due to the structural and chemical changes of aging, may be a less suitable substrate for molds compared with the two-week-old paddy.
Cost-benefit analysis of gamma irradiation for brown rice storage stability
Table 4 summarizes the estimated incremental costs (in USD) associated with irradiation, storage, and transportation of brown rice samples. The additional expenditures incurred per kilogram were US$ 0.02 for storage, at least US$ 0.14 for irradiation, and US$ 0.04 for transportation, relative to the prevailing market price of brown rice in the Philippines. These figures correspond to a 1.50, 10.53, and 3.01% increase, respectively. In comparison, the estimated cost of mitigating postharvest losses through conventional means amounted to US$ 0.40 per kilogram, reflecting a substantial 30.08% increase in total cost.
From an economic standpoint, while irradiation contributes to marginal increases in storage and transportation costs primarily due to extended shelf-life and logistical handling the overall financial impact is offset by the marked reduction in postharvest losses. Interestingly, based on the economic model used in this study, although initial investments in irradiation infrastructure and operations may appear significant, irradiation resulted in a 30% reduction in economic losses associated with spoilage and fungal contamination during storage of brown rice. Thus, the long-term cost savings due to reduced product loss substantiate the economic viability of food irradiation as a postharvest intervention strategy if scaled at a national level.
Table 3: Impact of irradiation, storage, and maturity on brown rice moisture content*
| Variety | Paddy age | Dose (kGy) | Packaging material | Moisture content (%) |
| SL-7 | 2 weeks | Control | SB | 10.11±0.42 ab |
| PE | 10.13±0.4 ab | |||
| 0.5 kGy | SB | 10.02±0.35 ab | ||
| PE | 10.15±0.39 ab | |||
| 1.0 kGy | SB | 9.82±0.3 ab | ||
| PE | 9.92±0.27 ab | |||
| 8 weeks | Control | SB | 10.24±1.03 ab | |
| PE | 10.22±1.18 b | |||
| 0.5 kGy | SB | 10.18±0.93 ab | ||
| PE | 10.09±0.9 ab | |||
| 1.0 kGy | SB | 10.04±1.11 ab | ||
| PE | 10.02±0.93 ab | |||
| RC-160 | 2 weeks | Control | SB | 9.29±0.55 ab |
| PE | 9.37±0.49 ab | |||
| 0.5 kGy | SB | 9.28±0.33 ab | ||
| PE | 9.4±0.39 ab | |||
| 1.0 kGy | SB | 9.19±0.47 a | ||
| PE | 9.34±0.4 ab | |||
| 8 weeks | Control | SB | 9.86±0.76 ab | |
| PE | 9.91±0.64 ab | |||
| 0.5 kGy | SB | 9.71±0.82 ab | ||
| PE | 9.76±0.75 ab | |||
| 1.0 kGy | SB | 9.68±0.65 ab | ||
| PE | 9.58±0.76 ab |
* Moisture content of brown rice (SL-7 and RC160 varieties) is presented for samples sourced from two-week-old and eight-week-old paddy based on the storage studies described in the Materials and Methods. Samples were either irradiated or non-irradiated and stored in IRRI Super Bag™ (SB) or standard Polyethylene (PE) bags. An ANOVA at α=0.05 assessed the effects of paddy age and packaging. Row-wise superscripts at each paddy age indicate a significant difference at the 95% confidence level.
Table 4: A cost-sensitivity analysis of gamma irradiation of brown rice
Table 4: A cost-sensitivity analysis of gamma irradiation of brown rice
| Additional cost (US$ per kg) | ||||
| Storage cost | Non-irradiated | Irradiated at 1 kGy | ||
| +0.00 | +0.02 (1.50%a) | |||
| Irradiation cost | Non-irradiated | 2015 Source activity | 2024 Source activity | |
| +0.00 | +0.63 (47.37%a) | +0.14 (10.53%a) | ||
| Transportation cost | Non-irradiated | Irradiated at 1 kGy | ||
| +0.00 | +0.04 (3.01%a) | |||
| Cost due to postharvest loss | Non-irradiated | Irradiated at 1 kGy | ||
| +0.00 | -0.40 (30.08%a) | |||
Beyond fungal control: perspectives on other benefits of brown rice irradiation
In food irradiation, a critical consideration alongside microbial safety is the impact on nutritional, antioxidant, and sensory properties of the treated product. Previous research by Gragasin et al. (2022) demonstrated that the nutritional profile of irradiated RC-160 brown rice remained stable even after eight months of storage, suggesting minimal degradation of key nutrients post-irradiation. Moreover, sensory evaluation revealed no significant differences in taste, odor, and texture between irradiated and non-irradiated samples, affirming that the overall acceptability of brown rice is not adversely affected by the irradiation process.
Beyond preservation, irradiation has also been shown to enhance certain functional qualities (Rita et al., 2024). A recent study by Silos et al. (2024) reported that gamma irradiation at 5 kGy significantly increased the total phenolic content of SL-218 brown rice, thereby boosting its antioxidant capacity. These findings underscore the dual potential of irradiation to not only ensure microbiological safety but also improve the nutritional and functional value of food commodities.
The efficacy of low-dose gamma irradiation in reducing fungal load in other cereals has been well established. For example, Aziz et al. (2006) demonstrated similar outcomes in various grains at 4 kGy. In rice and wheat, doses ranging from 3 to 5 kGy have been shown to inactivate key fungal genera such as Alternaria, Aspergillus, and Fusarium during extended storage periods of 6 to 12 months (Maity et al., 2009; Wang and Yu, 2010). Furthermore, Aziz et al. (2004) confirmed that gamma irradiation at 5 kGy effectively suppressed mold growth and mycotoxin production in maize, chickpeas, and groundnut seeds.
In addition to gamma irradiation, electron beam irradiation has emerged as an alternative modality with comparable efficacy. For instance, Sarrı́as et al. (2003) demonstrated that electron beam irradiation at 7.5 kGy using a 10-MeV circular accelerator successfully inactivated coliforms, Escherichia coli, Bacillus cereus, sulfite-reducing clostridia, and various fungi in both husked and unhusked rice. Similarly, electron beam treatment at 7.5 kGy has been reported to extend the shelf-life and improve the quality of brown and milled rice during long-term storage (Luo et al., 2019). Complementary results were obtained by Prakhongsil et al. (2025), who observed that exposure to 1 kGy from a 10-MeV electron beam significantly enhanced the functional properties of Thai Hom Mali germinated brown rice, increasing γ-aminobutyric acid content from 3.76 to 6.44 mg/100 g (dry weight), a 1.7-fold increase, while extending shelf stability.
Conclusion
This study confirms that gamma irradiation is an effective postharvest intervention for reducing fungal contamination and prolonging the shelf-life of brown rice under practical storage conditions. While the study successfully demonstrated the dose-dependent inactivation of A. niger and overall microbial reduction, a key limitation was the lack of direct measurement of mycotoxin levels, which are critical indicators of food safety. Future studies should investigate the efficacy of both gamma and electron beam irradiation in mitigating mycotoxin production across different fungal species and storage environments. Practically, the application of irradiation despite modest increases in processing costs offers a cost-effective strategy for reducing postharvest losses, improving food safety, and enhancing marketability of brown rice. These findings are particularly relevant for local farmers, food processors, and regulatory authorities seeking to implement scalable, science-based approaches to grain preservation and food security.
Author contributions
G.B.A., G.D., M.C.B.G., C.O.A., Z.M.D.G., and C.C.D. conceptualized the study; G.B.A., G.D., H.M.S., M.M.T., M.L.C.C., and C.C.D. developed the methodology; G.B.A. and G.D. handled software implementation and original draft writing; G.C. and G.B.A. carried out the formal analysis; M.M.T. and M.L.C.C. performed formal analysis; M.C.B.G., C.C.D., G.C., H.M.S., and C.O.A. contributed to manuscript writing-review; C.C.D. and C.O.A. edited the manuscript; M.C.B.G. provided resources; Z.M.D.G. and C.O.A. supervised the project; Z.M.D.G. managed project administration. C.C.D. conducted the visualization. All authors read and approved the final manuscript.
Acknowledgement
Maria Ysabel Domino is acknowledged for assistance in polishing the manuscript.
Conflicts of interest
The authors declare that there is no conflict of interest.
Funding
This research was funded by the Department of Agriculture-Philippine Center for Postharvest Development and Mechanization, Science City of Munoz, Nueva Ecija, Philippines and the DOST-Philippine Nuclear Research Institute GAA under the project “Sustainable Food Systems through Radiation Technology: Food Safety and Functionalization R&D”.
Ethical consideration
Not applicable.
References
Ali N. (2019). Aflatoxins in rice: worldwide occurrence and public health perspectives. Toxicology Reports. 6: 1188-1197. [DOI: 10.1016/j.toxrep.2019.11.007]
Asia Rice Foundation. (2018). The Philippine rice industry roadmap 2030. URL: https://www.philrice.gov.ph/wp-content/uploads/2018/09/The-Philippine-Rice-Industry-Roadmap-2030.pdf. Accessed 20 January 2024.
Association of Official Agricultural Chemists (AOAC). (2002). AOAC official method 997.02 yeast and mold counts in foods, dry rehydratable film method (Petrifilmtm method). URL: https://docslib.org/doc/10931992/aoac-997-02-yeast-and-mold-count-in-foods. Accessed 20 April 2025.
Aziz N.H., Mattar Z.A., Mahrous S.R. (2006). Contamination of grains by mycotoxin-producing molds and mycotoxins and control by gamma irradiation. Journal of Food Safety. 26: 184-201. [DOI: 10.1111/j.1745-4565.2006.00042.x]
Aziz N.H., Moussa L.A.A., Far F.M.E. (2004). Reduction of fungi and mycotoxins formation in seeds by gamma-radiation. Journal of Food Safety. 24: 109-127. [DOI: 10.1111/j.1745-4565.2004.tb00379.x]
Balendres M.A.O., Karlovsky P., Cumagun C.J.R. (2019). Mycotoxigenic fungi and mycotoxins in agricultural crop commodities in the Philippines: a review. Foods. 8: 249. [DOI: 10.3390/foods8070249 10.3390/foods8070249]
Boonchoo T., Jitareerat P., Photchanachai S., Chinaphuti A. (2005). Effect of gamma irradiation on Aspergillus flavus and brown rice quality during storage. Proceeding of the 2nd International Symposium on the New Frontiers of Irradiated Food and Non-Food Products, KMUTT, Bangkok, Thailand. 22-23.
Borba V.S.D., Paiva Rodrigues M.H., Badiale-Furlong E. (2020). Impact of biological contamination of rice on food safety. Food Reviews International, 36: 745-760. [DOI: 10.1080/87559129.2019.1683745]
Bureau of Agricultural and Fisheries Standards (BAFPS). (2014). Philippine national standard: code of good agricultural practices for rice, PNS BAFS 141:2014 ICS 65.020. URL: https://bafs.da.gov.ph/index.php/approved-philippine-national-standards/Accessed 12 February 2023.
Castro S.G. (2003). Postharvest technology in the Philippines. APCAEM Regional Seminar on Postharvest Technology for Major Crops. 153-162. URL: https://www.un-csam.org/sites/default/files/2021-01/Post-harvest% 20Technology%20in%20the%20Philippines.pdf.
Chrastil J. (1990). Chemical and physicochemical changes of rice during storage at different temperatures. Journal of Cereal Science. 11: 71-85. [DOI: 10.1016/S0733-5210(09)80182-3]
Cinar A., Onbaşı E. (2020). Mycotoxins: the hidden danger in foods. In: Sabuncuoğlu S. (Editor). Mycotoxins and food safety. IntechOpen, London. pp: 1-21. [DOI: 10.5772/intechopen.89001]
Cortésão M., De Haas A., Unterbusch R., Fujimori A., Schütze T., Meyer V., Moeller R. (2020). Aspergillus niger spores are highly resistant to space radiation. Frontiers in Microbiology. 11: 560-572. [DOI: 10.3389/fmicb.2020.00560]
De Guzman Z.M., De Dios C.C., Patindol J.A. (1998). Storage evaluation of gamma irradiated rice. IAEA International Nuclear Information System IAEA-CN-76/37P. URL: https://www.osti.gov/etdeweb/servlets/purl/20004653. Accessed 12 March 2025.
Eslami M., Mashak Z., Heshmati A., Shokrzadeh M., Mozaffari Nejad A.S. (2015). Determination of aflatoxin B1 levels in Iranian rice by ELISA method. Toxin Reviews. 34: 125-128. [DOI: 10.3109/15569543.2015.1074925]
Faruq G., Prodhan Z.H., Nezhadahmadi A. (2015). Effects of ageing on selected cooking quality parameters of rice. International Journal of Food Properties. 18: 922-933. [DOI: 10.1080/10942912.2014.913062]
Food and Agriculture Organization (FAO). (2004). FAO and climate change. URL: https://www.fao.org/3/i0144e/i0144e.pdf. Accessed 17 August 2023.
Glover D., Kim S.K., Stone G.D. (2020). Golden rice and technology adoption theory: a study of seed choice dynamics among rice growers in the Philippines. Technology in Society, 60: 101227. [DOI: 10.1016/j.techsoc.2019.101227]
Gragasin M.C.B., Villota S.M.M., Capariño O. A., De Leon A.A., Rustia J.M., Gantioque G.G., Juvinal J.G. (2022). Effect of gamma irradiation on the sensory quality of stored brown rice as influenced by age of paddy, packaging and storage period. Asian Journal of Postharvest and Mechanization. 5: 76-89. URL: https://www.philmech.gov.ph/resources/journal/AJPM%20Vol.%205%20No.1%202022_2.pdf. Accessed 12 March 2025.
Ijadpanahsaravi M., Snoek L.B., Teertstra W.R., Wösten H.A.B. (2024). The impact of inter-and intra-species spore density on germination of the food spoilage fungus Aspergillus niger. International Journal of Food Microbiology. 410: 110495. [DOI: 10.1016/j.ijfoodmicro.2023.110495]
International Rice Research Institute (IRRI). (2024). Paddy quality. URL: http://www.knowledgebank.irri.org/step-by-step-production/ postharvest/milling/milling-and-quality/item/paddy-quality. Accessed 01 January 2024.
International Rice Research Institute (IRRI). (2014). STAR: statistical tool for agricultural research, version 2.0. Los Baños, Philippines: IRRI. URL: https://www.irri.org/resources-and-tools/digital-tools. Accessed 12 March 2025.
Koch S.M., Freidank-Pohl C., Siontas O., Cortésão M., Mota A., Runzheimer K., Jung S., Rebrosova K., Siler M., Moeller R., Meyer V. (2023). Aspergillus niger as a cell factory for the production of pyomelanin, a molecule with UV-C radiation shielding activity. Frontiers in Microbiology. 14: 1233740. [DOI: 10.3389/fmicb.2023.1233740]
Kumar A., Khandai S., Singh M., Singh A., Panwar G.S., Kumar A., Singh S., Kumar V. (2020). Hermetic sealed storage IRRI super bag: reduces post-harvest losses in seed grains. International Journal of Chemical Studies. 8: 403-406. [DOI: 10.22271/chemi.2020.v8.i4g.10170]
Kushiro M. (2015). Historical review of researches on yellow rice and mycotoxigenic fungi adherent to rice in Japan. JSM Mycotoxins. 65:19-23. [DOI: 10.2520/myco.65.19]
Lee N.Y., Kim J.K. (2018). Effects of gamma radiation on the physicochemical properties of brown rice and changes in the quality of porridge. Radiation Physics and Chemistry. 152: 89-92. [DOI: 10.1016/j.radphyschem.2018.07.021]
Luo X., Li Y., Yang D., Xing J., Li K., Yang M., Wang R., Wang L., Zhang Y., Chen Z. (2019). Effects of electron beam irradiation on storability of brown and milled rice. Journal of Stored Products Research. 81: 22-30. [DOI: 10.1016/j.jspr.2018.12.003]
Mahajan P.V., Caleb O.J., Singh Z., Watkins C.B., Geyer M. (2014). Postharvest treatments of fresh produce. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences. 372: 20130309. [DOI: 10.1098/rsta.2013.0309]
Maity J.P., Kar S., Banerjee S., Chakraborty A., Santra S.C. (2009). Effects of gamma irradiation on long-storage seeds of Oryza sativa (cv. 2233) and their surface infecting fungal diversity. Radiation Physics and Chemistry. 78: 1106-1010. [DOI: 10.1016/j.radphyschem.2009.06.002]
National Food Authority (NFA). (2024). Warehouse library and status of warehouse utilization as of November 2021. URL: https://nfa.gov.ph/images/files/nfa_directory/nfa-warehouses-reg3.pdf. Accessed 12 January 2024.
Nemtanu M.R., Brasoveanu M., Grecu M.N., Minea R. (2005). Green coffee decontamination by electron beam irradiation. Nuclear Instruments and Methods in Physics Research Section B: Beam Interactions with Materials and Atoms. 240: 83-86. [DOI: 10.1016/j.nimb.2005.06.092]
Palomo A.M., Garcia R.G., Diaz D.A., Capanzana M.V. (2020). Optimization of moist-heat and dry-heat treatment for the production of stabilized brown rice using Box-Behnken design. Philippine Journal of Science. 149: 43-53. [DOI: 10.56899/149.01.05]
Parfitt J., Barthel M., Macnaughton S. (2010). Food waste within food supply chains: quantification and potential for change to 2050. Philosophical Transactions of the Royal Society B: Biological Sciences. 365: 3065-3081. [DOI: 10.1098/rstb.2010.0126]
Peng B., He L.L., Tan J., Zheng L.T., Zhang J.T., Qiao Q.W., Wang Y., Gao Y., Tian X.Y., Liu Z.Y., Song X.H., Sun Y.Y., et al. (2019). Effects of rice aging on its main nutrients and quality characters. Journal of Agricultural Science. 11: 44-56. [DOI: 10.5539/jas.v11n17p44]
Philippine Food and Drug Administration (Philippines-FDA). (2022). FDA circular No. 2022-012: guidelines on the microbiological requirements and assessment of certain prepackaged processed food products repealing FDA circular No. 2013-010 entitled “revised guidelines for the assessment of microbiological quality of processed foods”. URL: https://www.fda.gov.ph/fda-circular-no-2022-012-guidelines-on-the-microbiological-requirements-and-assessment-of-certain-prepackaged-processed-food-products-repealing-fda-circular-no-2013-010-entitled-revised-g/. Accessed 30 April 2025.
Philippine Rice Research Institute (PhilRice). (2018). NSIC Rc 160: tried and tested. URL: https://www.philrice.gov.ph/nsic-rc-160-tried-tested/. Accessed 12 January 2024.
Prakhongsil P., Sajjabut S., Pewlong W., Khemthong K., Eamsiri J., Picha R., Thamrongsiripak N. (2025). Increasing γ-aminobutyric acid in mixed germinated brown rice via electron beam irradiation. Trends in Sciences. 22: 8611. [DOI: 10.48048/tis.2024.8611]
Ramezanzadeh F.M., Rao R.M., Prinyawiwatkul W., Marshall W.E., Windhauser M. (2000). Effects of microwave heat, packaging, and storage temperature on fatty acid and proximate compositions in rice bran. Journal of Agricultural and Food Chemistry. 48: 464-467. [DOI: 10.1021/jf9909609]
Reddy K.R.N., Reddy C.S., Muralidharan K. (2009). Detection of Aspergillus spp. and aflatoxin B1 in rice in India. Food Microbiology. 26: 27-31. [DOI: 10.1016/j.fm.2008.07.013]
Rita K.M.M.S., Turla A.Y., Endriga M.A., Agapito J.D., Deocaris C.C. (2024). Beyond preservation: a systematic review on the impact of ionizing radiation on food functionality. Philippine Journal of Science. 153: 101-112.
Saikrishna A., Dutta S., Subramanian V., Moses J.A., Anandharamakrishnan C. (2018). Ageing of rice: a review. Journal of Cereal Science. 81: 161-170. [DOI: 10.1016/j.jcs.2018.04.009]
Saleh A.S., Wang P., Wang N., Yang L., Xiao Z. (2019). Brown rice versus white rice: nutritional quality, potential health benefits, development of food products, and preservation technologies. Comprehensive Reviews in Food Science and Food Safety. 18: 1070-1096. [DOI: 10.1111/1541-4337.12449]
Sales A.C., Yoshizawa T. (2005). Updated profile of aflatoxin and Aspergillus section Flavi contamination in rice and its byproducts from the Philippines. Food Additives and Contaminants. 22: 429-436. [DOI: 10.1080/02652030500058387]
Sarrı́as J.A., Valero M., Salmerón M.C. (2003). Elimination of Bacillus cereus contamination in raw rice by electron beam irradiation. Food Microbiology. 20: 327-332. [DOI: 10.1016/S0740-0020(02)00124-7]
Sharma A. (2004). Post-harvest processing of fruits and vegetables by ionizing radiation. Production practices and quality assessment of food crops: volume 4: preharvest treatment and technology. 1st edition. Springer Dordrecht, Netherlands. pp: 261-265. [DOI: 10.1007/1-4020-2535-1_10]
Shobha D., Vasudevan S.N., Badigannavar A. (2024). Impact of gamma irradiation and packaging on the storage quality of little millet (Panicum sumatrense) flour. Cereal Research Communications. 52: 777–787. [DOI: 10.1007/s42976-023-00391-5]
Silos A.P., Diano G.T., Abrera G.B., Baldos D.T., Gragasin M.C., Tolentino M.M., Deocaris C.C., Asaad C.O. (2024). Gamma irradiation as a method to enhance antioxidant activity and inhibit rancidification of brown rice. Radiation Physics and Chemistry, 223: 112026. [DOI: 10.1016/j.radphyschem.2024.112026]
Sorn V., Meas P., Pin T., Gummert M. (2017). Effects of drying and storage management on fungi (Aflatoxin B1) accumulation and rice quality in Cambodia. Journal of Agriculture and Rural Development in the Tropics and Subtropics. 118: 141-148.
Swamy Y.M.I., Sowbhagya C.M., Bhattacharya K.R. (1978). Changes in the physicochemical properties of rice with aging. Journal of the Science of Food and Agriculture. 29: 627-639. [DOI: 10.1002/jsfa.2740290709]
Tournas V., Stack M.E., Mislivec P.B., Koch H.A., Bandler, R. (2001). Chapter 18: yeasts, molds, and mycotoxins. In: Tournas V., Stack M.E., Mislivec P.B., Koch H.A., Bandler, R. (Editors). Bacteriological analytical manual (BAM). U.S. Food and Drug Administration, Maryland. URL: https://www.fda.gov/food/laboratory-methods-food/bam-chapter-18-yeasts-molds-and-mycotoxins.
Wang J., Yu Y. (2010). Inactivation of mildew in rough rice and wheat by gamma irradiation. Radiation Physics and Chemistry. 79: 740-743. [DOI: 10.1016/j.radphyschem.2009.12.001]
Wongnaa C.A., Ankomah E.D., Ojo T.O., Abokyi E., Sienso G., Awunyo-Vitor D. (2023). Valuing postharvest losses among tomato smallholder farmers: evidence from Ghana. Cogent Food and Agriculture. 9: 2187183. [DOI: 10.1080/23311932.2023.2187183]
Zhou Z., Robards K., Helliwell S., Blanchard C. (2002). Ageing of stored rice: changes in chemical and physical attributes. Journal of Cereal Science. 35: 65-78. [DOI: 10.1006/jcrs.2001.0418]
Zhou Z., Wang X., Si X., Blanchard C., Strappe P. (2015). The ageing mechanism of stored rice: a concept model from the past to the present. Journal of Stored Products Research. 64: 80-87. [DOI: 10.1016/j.jspr.2015.09.004]
In food irradiation, a critical consideration alongside microbial safety is the impact on nutritional, antioxidant, and sensory properties of the treated product. Previous research by Gragasin et al. (2022) demonstrated that the nutritional profile of irradiated RC-160 brown rice remained stable even after eight months of storage, suggesting minimal degradation of key nutrients post-irradiation. Moreover, sensory evaluation revealed no significant differences in taste, odor, and texture between irradiated and non-irradiated samples, affirming that the overall acceptability of brown rice is not adversely affected by the irradiation process.
Beyond preservation, irradiation has also been shown to enhance certain functional qualities (Rita et al., 2024). A recent study by Silos et al. (2024) reported that gamma irradiation at 5 kGy significantly increased the total phenolic content of SL-218 brown rice, thereby boosting its antioxidant capacity. These findings underscore the dual potential of irradiation to not only ensure microbiological safety but also improve the nutritional and functional value of food commodities.
The efficacy of low-dose gamma irradiation in reducing fungal load in other cereals has been well established. For example, Aziz et al. (2006) demonstrated similar outcomes in various grains at 4 kGy. In rice and wheat, doses ranging from 3 to 5 kGy have been shown to inactivate key fungal genera such as Alternaria, Aspergillus, and Fusarium during extended storage periods of 6 to 12 months (Maity et al., 2009; Wang and Yu, 2010). Furthermore, Aziz et al. (2004) confirmed that gamma irradiation at 5 kGy effectively suppressed mold growth and mycotoxin production in maize, chickpeas, and groundnut seeds.
In addition to gamma irradiation, electron beam irradiation has emerged as an alternative modality with comparable efficacy. For instance, Sarrı́as et al. (2003) demonstrated that electron beam irradiation at 7.5 kGy using a 10-MeV circular accelerator successfully inactivated coliforms, Escherichia coli, Bacillus cereus, sulfite-reducing clostridia, and various fungi in both husked and unhusked rice. Similarly, electron beam treatment at 7.5 kGy has been reported to extend the shelf-life and improve the quality of brown and milled rice during long-term storage (Luo et al., 2019). Complementary results were obtained by Prakhongsil et al. (2025), who observed that exposure to 1 kGy from a 10-MeV electron beam significantly enhanced the functional properties of Thai Hom Mali germinated brown rice, increasing γ-aminobutyric acid content from 3.76 to 6.44 mg/100 g (dry weight), a 1.7-fold increase, while extending shelf stability.
Conclusion
This study confirms that gamma irradiation is an effective postharvest intervention for reducing fungal contamination and prolonging the shelf-life of brown rice under practical storage conditions. While the study successfully demonstrated the dose-dependent inactivation of A. niger and overall microbial reduction, a key limitation was the lack of direct measurement of mycotoxin levels, which are critical indicators of food safety. Future studies should investigate the efficacy of both gamma and electron beam irradiation in mitigating mycotoxin production across different fungal species and storage environments. Practically, the application of irradiation despite modest increases in processing costs offers a cost-effective strategy for reducing postharvest losses, improving food safety, and enhancing marketability of brown rice. These findings are particularly relevant for local farmers, food processors, and regulatory authorities seeking to implement scalable, science-based approaches to grain preservation and food security.
Author contributions
G.B.A., G.D., M.C.B.G., C.O.A., Z.M.D.G., and C.C.D. conceptualized the study; G.B.A., G.D., H.M.S., M.M.T., M.L.C.C., and C.C.D. developed the methodology; G.B.A. and G.D. handled software implementation and original draft writing; G.C. and G.B.A. carried out the formal analysis; M.M.T. and M.L.C.C. performed formal analysis; M.C.B.G., C.C.D., G.C., H.M.S., and C.O.A. contributed to manuscript writing-review; C.C.D. and C.O.A. edited the manuscript; M.C.B.G. provided resources; Z.M.D.G. and C.O.A. supervised the project; Z.M.D.G. managed project administration. C.C.D. conducted the visualization. All authors read and approved the final manuscript.
Acknowledgement
Maria Ysabel Domino is acknowledged for assistance in polishing the manuscript.
Conflicts of interest
The authors declare that there is no conflict of interest.
Funding
This research was funded by the Department of Agriculture-Philippine Center for Postharvest Development and Mechanization, Science City of Munoz, Nueva Ecija, Philippines and the DOST-Philippine Nuclear Research Institute GAA under the project “Sustainable Food Systems through Radiation Technology: Food Safety and Functionalization R&D”.
Ethical consideration
Not applicable.
References
Ali N. (2019). Aflatoxins in rice: worldwide occurrence and public health perspectives. Toxicology Reports. 6: 1188-1197. [DOI: 10.1016/j.toxrep.2019.11.007]
Asia Rice Foundation. (2018). The Philippine rice industry roadmap 2030. URL: https://www.philrice.gov.ph/wp-content/uploads/2018/09/The-Philippine-Rice-Industry-Roadmap-2030.pdf. Accessed 20 January 2024.
Association of Official Agricultural Chemists (AOAC). (2002). AOAC official method 997.02 yeast and mold counts in foods, dry rehydratable film method (Petrifilmtm method). URL: https://docslib.org/doc/10931992/aoac-997-02-yeast-and-mold-count-in-foods. Accessed 20 April 2025.
Aziz N.H., Mattar Z.A., Mahrous S.R. (2006). Contamination of grains by mycotoxin-producing molds and mycotoxins and control by gamma irradiation. Journal of Food Safety. 26: 184-201. [DOI: 10.1111/j.1745-4565.2006.00042.x]
Aziz N.H., Moussa L.A.A., Far F.M.E. (2004). Reduction of fungi and mycotoxins formation in seeds by gamma-radiation. Journal of Food Safety. 24: 109-127. [DOI: 10.1111/j.1745-4565.2004.tb00379.x]
Balendres M.A.O., Karlovsky P., Cumagun C.J.R. (2019). Mycotoxigenic fungi and mycotoxins in agricultural crop commodities in the Philippines: a review. Foods. 8: 249. [DOI: 10.3390/foods8070249 10.3390/foods8070249]
Boonchoo T., Jitareerat P., Photchanachai S., Chinaphuti A. (2005). Effect of gamma irradiation on Aspergillus flavus and brown rice quality during storage. Proceeding of the 2nd International Symposium on the New Frontiers of Irradiated Food and Non-Food Products, KMUTT, Bangkok, Thailand. 22-23.
Borba V.S.D., Paiva Rodrigues M.H., Badiale-Furlong E. (2020). Impact of biological contamination of rice on food safety. Food Reviews International, 36: 745-760. [DOI: 10.1080/87559129.2019.1683745]
Bureau of Agricultural and Fisheries Standards (BAFPS). (2014). Philippine national standard: code of good agricultural practices for rice, PNS BAFS 141:2014 ICS 65.020. URL: https://bafs.da.gov.ph/index.php/approved-philippine-national-standards/Accessed 12 February 2023.
Castro S.G. (2003). Postharvest technology in the Philippines. APCAEM Regional Seminar on Postharvest Technology for Major Crops. 153-162. URL: https://www.un-csam.org/sites/default/files/2021-01/Post-harvest% 20Technology%20in%20the%20Philippines.pdf.
Chrastil J. (1990). Chemical and physicochemical changes of rice during storage at different temperatures. Journal of Cereal Science. 11: 71-85. [DOI: 10.1016/S0733-5210(09)80182-3]
Cinar A., Onbaşı E. (2020). Mycotoxins: the hidden danger in foods. In: Sabuncuoğlu S. (Editor). Mycotoxins and food safety. IntechOpen, London. pp: 1-21. [DOI: 10.5772/intechopen.89001]
Cortésão M., De Haas A., Unterbusch R., Fujimori A., Schütze T., Meyer V., Moeller R. (2020). Aspergillus niger spores are highly resistant to space radiation. Frontiers in Microbiology. 11: 560-572. [DOI: 10.3389/fmicb.2020.00560]
De Guzman Z.M., De Dios C.C., Patindol J.A. (1998). Storage evaluation of gamma irradiated rice. IAEA International Nuclear Information System IAEA-CN-76/37P. URL: https://www.osti.gov/etdeweb/servlets/purl/20004653. Accessed 12 March 2025.
Eslami M., Mashak Z., Heshmati A., Shokrzadeh M., Mozaffari Nejad A.S. (2015). Determination of aflatoxin B1 levels in Iranian rice by ELISA method. Toxin Reviews. 34: 125-128. [DOI: 10.3109/15569543.2015.1074925]
Faruq G., Prodhan Z.H., Nezhadahmadi A. (2015). Effects of ageing on selected cooking quality parameters of rice. International Journal of Food Properties. 18: 922-933. [DOI: 10.1080/10942912.2014.913062]
Food and Agriculture Organization (FAO). (2004). FAO and climate change. URL: https://www.fao.org/3/i0144e/i0144e.pdf. Accessed 17 August 2023.
Glover D., Kim S.K., Stone G.D. (2020). Golden rice and technology adoption theory: a study of seed choice dynamics among rice growers in the Philippines. Technology in Society, 60: 101227. [DOI: 10.1016/j.techsoc.2019.101227]
Gragasin M.C.B., Villota S.M.M., Capariño O. A., De Leon A.A., Rustia J.M., Gantioque G.G., Juvinal J.G. (2022). Effect of gamma irradiation on the sensory quality of stored brown rice as influenced by age of paddy, packaging and storage period. Asian Journal of Postharvest and Mechanization. 5: 76-89. URL: https://www.philmech.gov.ph/resources/journal/AJPM%20Vol.%205%20No.1%202022_2.pdf. Accessed 12 March 2025.
Ijadpanahsaravi M., Snoek L.B., Teertstra W.R., Wösten H.A.B. (2024). The impact of inter-and intra-species spore density on germination of the food spoilage fungus Aspergillus niger. International Journal of Food Microbiology. 410: 110495. [DOI: 10.1016/j.ijfoodmicro.2023.110495]
International Rice Research Institute (IRRI). (2024). Paddy quality. URL: http://www.knowledgebank.irri.org/step-by-step-production/ postharvest/milling/milling-and-quality/item/paddy-quality. Accessed 01 January 2024.
International Rice Research Institute (IRRI). (2014). STAR: statistical tool for agricultural research, version 2.0. Los Baños, Philippines: IRRI. URL: https://www.irri.org/resources-and-tools/digital-tools. Accessed 12 March 2025.
Koch S.M., Freidank-Pohl C., Siontas O., Cortésão M., Mota A., Runzheimer K., Jung S., Rebrosova K., Siler M., Moeller R., Meyer V. (2023). Aspergillus niger as a cell factory for the production of pyomelanin, a molecule with UV-C radiation shielding activity. Frontiers in Microbiology. 14: 1233740. [DOI: 10.3389/fmicb.2023.1233740]
Kumar A., Khandai S., Singh M., Singh A., Panwar G.S., Kumar A., Singh S., Kumar V. (2020). Hermetic sealed storage IRRI super bag: reduces post-harvest losses in seed grains. International Journal of Chemical Studies. 8: 403-406. [DOI: 10.22271/chemi.2020.v8.i4g.10170]
Kushiro M. (2015). Historical review of researches on yellow rice and mycotoxigenic fungi adherent to rice in Japan. JSM Mycotoxins. 65:19-23. [DOI: 10.2520/myco.65.19]
Lee N.Y., Kim J.K. (2018). Effects of gamma radiation on the physicochemical properties of brown rice and changes in the quality of porridge. Radiation Physics and Chemistry. 152: 89-92. [DOI: 10.1016/j.radphyschem.2018.07.021]
Luo X., Li Y., Yang D., Xing J., Li K., Yang M., Wang R., Wang L., Zhang Y., Chen Z. (2019). Effects of electron beam irradiation on storability of brown and milled rice. Journal of Stored Products Research. 81: 22-30. [DOI: 10.1016/j.jspr.2018.12.003]
Mahajan P.V., Caleb O.J., Singh Z., Watkins C.B., Geyer M. (2014). Postharvest treatments of fresh produce. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences. 372: 20130309. [DOI: 10.1098/rsta.2013.0309]
Maity J.P., Kar S., Banerjee S., Chakraborty A., Santra S.C. (2009). Effects of gamma irradiation on long-storage seeds of Oryza sativa (cv. 2233) and their surface infecting fungal diversity. Radiation Physics and Chemistry. 78: 1106-1010. [DOI: 10.1016/j.radphyschem.2009.06.002]
National Food Authority (NFA). (2024). Warehouse library and status of warehouse utilization as of November 2021. URL: https://nfa.gov.ph/images/files/nfa_directory/nfa-warehouses-reg3.pdf. Accessed 12 January 2024.
Nemtanu M.R., Brasoveanu M., Grecu M.N., Minea R. (2005). Green coffee decontamination by electron beam irradiation. Nuclear Instruments and Methods in Physics Research Section B: Beam Interactions with Materials and Atoms. 240: 83-86. [DOI: 10.1016/j.nimb.2005.06.092]
Palomo A.M., Garcia R.G., Diaz D.A., Capanzana M.V. (2020). Optimization of moist-heat and dry-heat treatment for the production of stabilized brown rice using Box-Behnken design. Philippine Journal of Science. 149: 43-53. [DOI: 10.56899/149.01.05]
Parfitt J., Barthel M., Macnaughton S. (2010). Food waste within food supply chains: quantification and potential for change to 2050. Philosophical Transactions of the Royal Society B: Biological Sciences. 365: 3065-3081. [DOI: 10.1098/rstb.2010.0126]
Peng B., He L.L., Tan J., Zheng L.T., Zhang J.T., Qiao Q.W., Wang Y., Gao Y., Tian X.Y., Liu Z.Y., Song X.H., Sun Y.Y., et al. (2019). Effects of rice aging on its main nutrients and quality characters. Journal of Agricultural Science. 11: 44-56. [DOI: 10.5539/jas.v11n17p44]
Philippine Food and Drug Administration (Philippines-FDA). (2022). FDA circular No. 2022-012: guidelines on the microbiological requirements and assessment of certain prepackaged processed food products repealing FDA circular No. 2013-010 entitled “revised guidelines for the assessment of microbiological quality of processed foods”. URL: https://www.fda.gov.ph/fda-circular-no-2022-012-guidelines-on-the-microbiological-requirements-and-assessment-of-certain-prepackaged-processed-food-products-repealing-fda-circular-no-2013-010-entitled-revised-g/. Accessed 30 April 2025.
Philippine Rice Research Institute (PhilRice). (2018). NSIC Rc 160: tried and tested. URL: https://www.philrice.gov.ph/nsic-rc-160-tried-tested/. Accessed 12 January 2024.
Prakhongsil P., Sajjabut S., Pewlong W., Khemthong K., Eamsiri J., Picha R., Thamrongsiripak N. (2025). Increasing γ-aminobutyric acid in mixed germinated brown rice via electron beam irradiation. Trends in Sciences. 22: 8611. [DOI: 10.48048/tis.2024.8611]
Ramezanzadeh F.M., Rao R.M., Prinyawiwatkul W., Marshall W.E., Windhauser M. (2000). Effects of microwave heat, packaging, and storage temperature on fatty acid and proximate compositions in rice bran. Journal of Agricultural and Food Chemistry. 48: 464-467. [DOI: 10.1021/jf9909609]
Reddy K.R.N., Reddy C.S., Muralidharan K. (2009). Detection of Aspergillus spp. and aflatoxin B1 in rice in India. Food Microbiology. 26: 27-31. [DOI: 10.1016/j.fm.2008.07.013]
Rita K.M.M.S., Turla A.Y., Endriga M.A., Agapito J.D., Deocaris C.C. (2024). Beyond preservation: a systematic review on the impact of ionizing radiation on food functionality. Philippine Journal of Science. 153: 101-112.
Saikrishna A., Dutta S., Subramanian V., Moses J.A., Anandharamakrishnan C. (2018). Ageing of rice: a review. Journal of Cereal Science. 81: 161-170. [DOI: 10.1016/j.jcs.2018.04.009]
Saleh A.S., Wang P., Wang N., Yang L., Xiao Z. (2019). Brown rice versus white rice: nutritional quality, potential health benefits, development of food products, and preservation technologies. Comprehensive Reviews in Food Science and Food Safety. 18: 1070-1096. [DOI: 10.1111/1541-4337.12449]
Sales A.C., Yoshizawa T. (2005). Updated profile of aflatoxin and Aspergillus section Flavi contamination in rice and its byproducts from the Philippines. Food Additives and Contaminants. 22: 429-436. [DOI: 10.1080/02652030500058387]
Sarrı́as J.A., Valero M., Salmerón M.C. (2003). Elimination of Bacillus cereus contamination in raw rice by electron beam irradiation. Food Microbiology. 20: 327-332. [DOI: 10.1016/S0740-0020(02)00124-7]
Sharma A. (2004). Post-harvest processing of fruits and vegetables by ionizing radiation. Production practices and quality assessment of food crops: volume 4: preharvest treatment and technology. 1st edition. Springer Dordrecht, Netherlands. pp: 261-265. [DOI: 10.1007/1-4020-2535-1_10]
Shobha D., Vasudevan S.N., Badigannavar A. (2024). Impact of gamma irradiation and packaging on the storage quality of little millet (Panicum sumatrense) flour. Cereal Research Communications. 52: 777–787. [DOI: 10.1007/s42976-023-00391-5]
Silos A.P., Diano G.T., Abrera G.B., Baldos D.T., Gragasin M.C., Tolentino M.M., Deocaris C.C., Asaad C.O. (2024). Gamma irradiation as a method to enhance antioxidant activity and inhibit rancidification of brown rice. Radiation Physics and Chemistry, 223: 112026. [DOI: 10.1016/j.radphyschem.2024.112026]
Sorn V., Meas P., Pin T., Gummert M. (2017). Effects of drying and storage management on fungi (Aflatoxin B1) accumulation and rice quality in Cambodia. Journal of Agriculture and Rural Development in the Tropics and Subtropics. 118: 141-148.
Swamy Y.M.I., Sowbhagya C.M., Bhattacharya K.R. (1978). Changes in the physicochemical properties of rice with aging. Journal of the Science of Food and Agriculture. 29: 627-639. [DOI: 10.1002/jsfa.2740290709]
Tournas V., Stack M.E., Mislivec P.B., Koch H.A., Bandler, R. (2001). Chapter 18: yeasts, molds, and mycotoxins. In: Tournas V., Stack M.E., Mislivec P.B., Koch H.A., Bandler, R. (Editors). Bacteriological analytical manual (BAM). U.S. Food and Drug Administration, Maryland. URL: https://www.fda.gov/food/laboratory-methods-food/bam-chapter-18-yeasts-molds-and-mycotoxins.
Wang J., Yu Y. (2010). Inactivation of mildew in rough rice and wheat by gamma irradiation. Radiation Physics and Chemistry. 79: 740-743. [DOI: 10.1016/j.radphyschem.2009.12.001]
Wongnaa C.A., Ankomah E.D., Ojo T.O., Abokyi E., Sienso G., Awunyo-Vitor D. (2023). Valuing postharvest losses among tomato smallholder farmers: evidence from Ghana. Cogent Food and Agriculture. 9: 2187183. [DOI: 10.1080/23311932.2023.2187183]
Zhou Z., Robards K., Helliwell S., Blanchard C. (2002). Ageing of stored rice: changes in chemical and physical attributes. Journal of Cereal Science. 35: 65-78. [DOI: 10.1006/jcrs.2001.0418]
Zhou Z., Wang X., Si X., Blanchard C., Strappe P. (2015). The ageing mechanism of stored rice: a concept model from the past to the present. Journal of Stored Products Research. 64: 80-87. [DOI: 10.1016/j.jspr.2015.09.004]
[*] Corresponding author (C.C. Deocaris)
* E-mail: ccdeocaris@pnri.dost.gov.ph
ORCID ID: https://orcid.org/0000-0003-3530-1228
* E-mail: ccdeocaris@pnri.dost.gov.ph
ORCID ID: https://orcid.org/0000-0003-3530-1228
Type of Study: Original article |
Subject:
Special
Received: 24/11/27 | Accepted: 25/04/30 | Published: 25/06/22
Received: 24/11/27 | Accepted: 25/04/30 | Published: 25/06/22
References
1. Ali N. (2019). Aflatoxins in rice: worldwide occurrence and public health perspectives. Toxicology Reports. 6: 1188-1197. [DOI: 10.1016/j.toxrep.2019.11.007] [DOI:10.1016/j.toxrep.2019.11.007] [PMID] [PMCID]
2. Asia Rice Foundation. (2018). The Philippine rice industry roadmap 2030. URL: https://www.philrice.gov.ph/wp-content/uploads/2018/09/The-Philippine-Rice-Industry-Roadmap-2030.pdf. Accessed 20 January 2024.
3. Association of Official Agricultural Chemists (AOAC). (2002). AOAC official method 997.02 yeast and mold counts in foods, dry rehydratable film method (Petrifilmtm method). URL: https://docslib.org/doc/10931992/aoac-997-02-yeast-and-mold-count-in-foods. Accessed 20 April 2025.
4. Aziz N.H., Mattar Z.A., Mahrous S.R. (2006). Contamination of grains by mycotoxin-producing molds and mycotoxins and control by gamma irradiation. Journal of Food Safety. 26: 184-201. [DOI: 10.1111/j.1745-4565.2006.00042.x] [DOI:10.1111/j.1745-4565.2006.00042.x]
5. Aziz N.H., Moussa L.A.A., Far F.M.E. (2004). Reduction of fungi and mycotoxins formation in seeds by gamma-radiation. Journal of Food Safety. 24: 109-127. [DOI: 10.1111/j.1745-4565.2004.tb00379.x] [DOI:10.1111/j.1745-4565.2004.tb00379.x]
6. Balendres M.A.O., Karlovsky P., Cumagun C.J.R. (2019). Mycotoxigenic fungi and mycotoxins in agricultural crop commodities in the Philippines: a review. Foods. 8: 249. [DOI: 10.3390/foods8070249 10.3390/foods8070249] [DOI:10.3390/foods8070249] [PMID] [PMCID]
7. Boonchoo T., Jitareerat P., Photchanachai S., Chinaphuti A. (2005). Effect of gamma irradiation on Aspergillus flavus and brown rice quality during storage. Proceeding of the 2nd International Symposium on the New Frontiers of Irradiated Food and Non-Food Products, KMUTT, Bangkok, Thailand. 22-23.
8. Borba V.S.D., Paiva Rodrigues M.H., Badiale-Furlong E. (2020). Impact of biological contamination of rice on food safety. Food Reviews International, 36: 745-760. [DOI: 10.1080/87559129.2019.1683745] [DOI:10.1080/87559129.2019.1683745]
9. Bureau of Agricultural and Fisheries Standards (BAFPS). (2014). Philippine national standard: code of good agricultural practices for rice, PNS BAFS 141:2014 ICS 65.020. URL: https://bafs.da.gov.ph/index.php/approved-philippine-national-standards/Accessed 12 February 2023.
10. Castro S.G. (2003). Postharvest technology in the Philippines. APCAEM Regional Seminar on Postharvest Technology for Major Crops. 153-162. URL: https://www.un-csam.org/sites/default/files/2021-01/Post-harvest% 20Technology%20in%20the%20Philippines.pdf.
11. Chrastil J. (1990). Chemical and physicochemical changes of rice during storage at different temperatures. Journal of Cereal Science. 11: 71-85. [DOI: 10.1016/S0733-5210(09)80182-3] [DOI:10.1016/S0733-5210(09)80182-3]
12. Cinar A., Onbaşı E. (2020). Mycotoxins: the hidden danger in foods. In: Sabuncuoğlu S. (Editor). Mycotoxins and food safety. IntechOpen, London. pp: 1-21. [DOI: 10.5772/intechopen.89001] [DOI:10.5772/intechopen.89001]
13. Cortésão M., De Haas A., Unterbusch R., Fujimori A., Schütze T., Meyer V., Moeller R. (2020). Aspergillus niger spores are highly resistant to space radiation. Frontiers in Microbiology. 11: 560-572. [DOI: 10.3389/fmicb.2020.00560] [DOI:10.3389/fmicb.2020.00560] [PMID] [PMCID]
14. De Guzman Z.M., De Dios C.C., Patindol J.A. (1998). Storage evaluation of gamma irradiated rice. IAEA International Nuclear Information System IAEA-CN-76/37P. URL: https://www.osti.gov/etdeweb/servlets/purl/20004653. Accessed 12 March 2025.
15. Eslami M., Mashak Z., Heshmati A., Shokrzadeh M., Mozaffari Nejad A.S. (2015). Determination of aflatoxin B1 levels in Iranian rice by ELISA method. Toxin Reviews. 34: 125-128. [DOI: 10.3109/15569543.2015.1074925] [DOI:10.3109/15569543.2015.1074925]
16. Faruq G., Prodhan Z.H., Nezhadahmadi A. (2015). Effects of ageing on selected cooking quality parameters of rice. International Journal of Food Properties. 18: 922-933. [DOI: 10.1080/10942912.2014.913062] [DOI:10.1080/10942912.2014.913062]
17. Food and Agriculture Organization (FAO). (2004). FAO and climate change. URL: https://www.fao.org/3/i0144e/i0144e.pdf. Accessed 17 August 2023.
18. Glover D., Kim S.K., Stone G.D. (2020). Golden rice and technology adoption theory: a study of seed choice dynamics among rice growers in the Philippines. Technology in Society, 60: 101227. [DOI: 10.1016/j.techsoc.2019.101227] [DOI:10.1016/j.techsoc.2019.101227]
19. Gragasin M.C.B., Villota S.M.M., Capariño O. A., De Leon A.A., Rustia J.M., Gantioque G.G., Juvinal J.G. (2022). Effect of gamma irradiation on the sensory quality of stored brown rice as influenced by age of paddy, packaging and storage period. Asian Journal of Postharvest and Mechanization. 5: 76-89. URL: https://www.philmech.gov.ph/resources/journal/AJPM%20Vol.%205%20No.1%202022_2.pdf. Accessed 12 March 2025.
20. Ijadpanahsaravi M., Snoek L.B., Teertstra W.R., Wösten H.A.B. (2024). The impact of inter-and intra-species spore density on germination of the food spoilage fungus Aspergillus niger. International Journal of Food Microbiology. 410: 110495. [DOI: 10.1016/j.ijfoodmicro.2023.110495] [DOI:10.1016/j.ijfoodmicro.2023.110495] [PMID]
21. International Rice Research Institute (IRRI). (2024). Paddy quality. URL: http://www.knowledgebank.irri.org/step-by-step-production/ postharvest/milling/milling-and-quality/item/paddy-quality. Accessed 01 January 2024.
22. International Rice Research Institute (IRRI). (2014). STAR: statistical tool for agricultural research, version 2.0. Los Baños, Philippines: IRRI. URL: https://www.irri.org/resources-and-tools/digital-tools. Accessed 12 March 2025.
23. Koch S.M., Freidank-Pohl C., Siontas O., Cortésão M., Mota A., Runzheimer K., Jung S., Rebrosova K., Siler M., Moeller R., Meyer V. (2023). Aspergillus niger as a cell factory for the production of pyomelanin, a molecule with UV-C radiation shielding activity. Frontiers in Microbiology. 14: 1233740. [DOI: 10.3389/fmicb.2023.1233740] [DOI:10.3389/fmicb.2023.1233740] [PMID] [PMCID]
24. Kumar A., Khandai S., Singh M., Singh A., Panwar G.S., Kumar A., Singh S., Kumar V. (2020). Hermetic sealed storage IRRI super bag: reduces post-harvest losses in seed grains. International Journal of Chemical Studies. 8: 403-406. [DOI: 10.22271/chemi.2020.v8.i4g.10170] [DOI:10.22271/chemi.2020.v8.i4g.10170]
25. Kushiro M. (2015). Historical review of researches on yellow rice and mycotoxigenic fungi adherent to rice in Japan. JSM Mycotoxins. 65:19-23. [DOI: 10.2520/myco.65.19] [DOI:10.2520/myco.65.19]
26. Lee N.Y., Kim J.K. (2018). Effects of gamma radiation on the physicochemical properties of brown rice and changes in the quality of porridge. Radiation Physics and Chemistry. 152: 89-92. [DOI: 10.1016/j.radphyschem.2018.07.021] [DOI:10.1016/j.radphyschem.2018.07.021]
27. Luo X., Li Y., Yang D., Xing J., Li K., Yang M., Wang R., Wang L., Zhang Y., Chen Z. (2019). Effects of electron beam irradiation on storability of brown and milled rice. Journal of Stored Products Research. 81: 22-30. [DOI: 10.1016/j.jspr.2018.12.003] [DOI:10.1016/j.jspr.2018.12.003]
28. Mahajan P.V., Caleb O.J., Singh Z., Watkins C.B., Geyer M. (2014). Postharvest treatments of fresh produce. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences. 372: 20130309. [DOI: 10.1098/rsta.2013.0309] [DOI:10.1098/rsta.2013.0309] [PMID] [PMCID]
29. Maity J.P., Kar S., Banerjee S., Chakraborty A., Santra S.C. (2009). Effects of gamma irradiation on long-storage seeds of Oryza sativa (cv. 2233) and their surface infecting fungal diversity. Radiation Physics and Chemistry. 78: 1106-1010. [DOI: 10.1016/j.radphyschem.2009.06.002] [DOI:10.1016/j.radphyschem.2009.06.002]
30. National Food Authority (NFA). (2024). Warehouse library and status of warehouse utilization as of November 2021. URL: https://nfa.gov.ph/images/files/nfa_directory/nfa-warehouses-reg3.pdf. Accessed 12 January 2024.
31. Nemtanu M.R., Brasoveanu M., Grecu M.N., Minea R. (2005). Green coffee decontamination by electron beam irradiation. Nuclear Instruments and Methods in Physics Research Section B: Beam Interactions with Materials and Atoms. 240: 83-86. [DOI: 10.1016/j.nimb.2005.06.092] [DOI:10.1016/j.nimb.2005.06.092]
32. Palomo A.M., Garcia R.G., Diaz D.A., Capanzana M.V. (2020). Optimization of moist-heat and dry-heat treatment for the production of stabilized brown rice using Box-Behnken design. Philippine Journal of Science. 149: 43-53. [DOI: 10.56899/149.01.05] [DOI:10.56899/149.01.05]
33. Parfitt J., Barthel M., Macnaughton S. (2010). Food waste within food supply chains: quantification and potential for change to 2050. Philosophical Transactions of the Royal Society B: Biological Sciences. 365: 3065-3081. [DOI: 10.1098/rstb.2010.0126] [DOI:10.1098/rstb.2010.0126] [PMID] [PMCID]
34. Peng B., He L.L., Tan J., Zheng L.T., Zhang J.T., Qiao Q.W., Wang Y., Gao Y., Tian X.Y., Liu Z.Y., Song X.H., Sun Y.Y., et al. (2019). Effects of rice aging on its main nutrients and quality characters. Journal of Agricultural Science. 11: 44-56. [DOI: 10.5539/jas.v11n17p44] [DOI:10.5539/jas.v11n17p44]
35. Philippine Food and Drug Administration (Philippines-FDA). (2022). FDA circular No. 2022-012: guidelines on the microbiological requirements and assessment of certain prepackaged processed food products repealing FDA circular No. 2013-010 entitled "revised guidelines for the assessment of microbiological quality of processed foods". URL: https://www.fda.gov.ph/fda-circular-no-2022-012-guidelines-on-the-microbiological-requirements-and-assessment-of-certain-prepackaged-processed-food-products-repealing-fda-circular-no-2013-010-entitled-revised-g/. Accessed 30 April 2025.
36. Philippine Rice Research Institute (PhilRice). (2018). NSIC Rc 160: tried and tested. URL: https://www.philrice.gov.ph/nsic-rc-160-tried-tested/. Accessed 12 January 2024.
37. Prakhongsil P., Sajjabut S., Pewlong W., Khemthong K., Eamsiri J., Picha R., Thamrongsiripak N. (2025). Increasing γ-aminobutyric acid in mixed germinated brown rice via electron beam irradiation. Trends in Sciences. 22: 8611. [DOI: 10.48048/tis.2024.8611] [DOI:10.48048/tis.2024.8611]
38. Ramezanzadeh F.M., Rao R.M., Prinyawiwatkul W., Marshall W.E., Windhauser M. (2000). Effects of microwave heat, packaging, and storage temperature on fatty acid and proximate compositions in rice bran. Journal of Agricultural and Food Chemistry. 48: 464-467. [DOI: 10.1021/jf9909609] [DOI:10.1021/jf9909609] [PMID]
39. Reddy K.R.N., Reddy C.S., Muralidharan K. (2009). Detection of Aspergillus spp. and aflatoxin B1 in rice in India. Food Microbiology. 26: 27-31. [DOI: 10.1016/j.fm.2008.07.013] [DOI:10.1016/j.fm.2008.07.013] [PMID]
40. Rita K.M.M.S., Turla A.Y., Endriga M.A., Agapito J.D., Deocaris C.C. (2024). Beyond preservation: a systematic review on the impact of ionizing radiation on food functionality. Philippine Journal of Science. 153: 101-112.
41. Saikrishna A., Dutta S., Subramanian V., Moses J.A., Anandharamakrishnan C. (2018). Ageing of rice: a review. Journal of Cereal Science. 81: 161-170. [DOI: 10.1016/j.jcs.2018.04.009] [DOI:10.1016/j.jcs.2018.04.009]
42. Saleh A.S., Wang P., Wang N., Yang L., Xiao Z. (2019). Brown rice versus white rice: nutritional quality, potential health benefits, development of food products, and preservation technologies. Comprehensive Reviews in Food Science and Food Safety. 18: 1070-1096. [DOI: 10.1111/1541-4337.12449] [DOI:10.1111/1541-4337.12449] [PMID]
43. Sales A.C., Yoshizawa T. (2005). Updated profile of aflatoxin and Aspergillus section Flavi contamination in rice and its byproducts from the Philippines. Food Additives and Contaminants. 22: 429-436. [DOI: 10.1080/02652030500058387] [DOI:10.1080/02652030500058387] [PMID]
44. Sarrı́as J.A., Valero M., Salmerón M.C. (2003). Elimination of Bacillus cereus contamination in raw rice by electron beam irradiation. Food Microbiology. 20: 327-332. [DOI: 10.1016/S0740-0020(02)00124-7] [DOI:10.1016/S0740-0020(02)00124-7]
45. Sharma A. (2004). Post-harvest processing of fruits and vegetables by ionizing radiation. Production practices and quality assessment of food crops: volume 4: preharvest treatment and technology. 1st edition. Springer Dordrecht, Netherlands. pp: 261-265. [DOI: 10.1007/1-4020-2535-1_10] [DOI:10.1007/1-4020-2535-1_10] [PMID]
46. Shobha D., Vasudevan S.N., Badigannavar A. (2024). Impact of gamma irradiation and packaging on the storage quality of little millet (Panicum sumatrense) flour. Cereal Research Communications. 52: 777-787. [DOI: 10.1007/s42976-023-00391-5] [DOI:10.1007/s42976-023-00391-5]
47. Silos A.P., Diano G.T., Abrera G.B., Baldos D.T., Gragasin M.C., Tolentino M.M., Deocaris C.C., Asaad C.O. (2024). Gamma irradiation as a method to enhance antioxidant activity and inhibit rancidification of brown rice. Radiation Physics and Chemistry, 223: 112026. [DOI: 10.1016/j.radphyschem.2024.112026] [DOI:10.1016/j.radphyschem.2024.112026]
48. Sorn V., Meas P., Pin T., Gummert M. (2017). Effects of drying and storage management on fungi (Aflatoxin B1) accumulation and rice quality in Cambodia. Journal of Agriculture and Rural Development in the Tropics and Subtropics. 118: 141-148.
49. Swamy Y.M.I., Sowbhagya C.M., Bhattacharya K.R. (1978). Changes in the physicochemical properties of rice with aging. Journal of the Science of Food and Agriculture. 29: 627-639. [DOI: 10.1002/jsfa.2740290709] [DOI:10.1002/jsfa.2740290709]
50. Tournas V., Stack M.E., Mislivec P.B., Koch H.A., Bandler, R. (2001). Chapter 18: yeasts, molds, and mycotoxins. In: Tournas V., Stack M.E., Mislivec P.B., Koch H.A., Bandler, R. (Editors). Bacteriological analytical manual (BAM). U.S. Food and Drug Administration, Maryland. URL: https://www.fda.gov/food/laboratory-methods-food/bam-chapter-18-yeasts-molds-and-mycotoxins.
51. Wang J., Yu Y. (2010). Inactivation of mildew in rough rice and wheat by gamma irradiation. Radiation Physics and Chemistry. 79: 740-743. [DOI: 10.1016/j.radphyschem.2009.12.001] [DOI:10.1016/j.radphyschem.2009.12.001]
52. Wongnaa C.A., Ankomah E.D., Ojo T.O., Abokyi E., Sienso G., Awunyo-Vitor D. (2023). Valuing postharvest losses among tomato smallholder farmers: evidence from Ghana. Cogent Food and Agriculture. 9: 2187183. [DOI: 10.1080/23311932.2023.2187183] [DOI:10.1080/23311932.2023.2187183]
53. Zhou Z., Robards K., Helliwell S., Blanchard C. (2002). Ageing of stored rice: changes in chemical and physical attributes. Journal of Cereal Science. 35: 65-78. [DOI: 10.1006/jcrs.2001.0418] [DOI:10.1006/jcrs.2001.0418]
54. Zhou Z., Wang X., Si X., Blanchard C., Strappe P. (2015). The ageing mechanism of stored rice: a concept model from the past to the present. Journal of Stored Products Research. 64: 80-87. [DOI: 10.1016/j.jspr.2015.09.004] [DOI:10.1016/j.jspr.2015.09.004]
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |




